Anionic amino acid surfactants (AAS), the most dominant and researched category among amino acid surfactants, hold the largest market share. Like other anionic surfactants, anionic amino acid surfactants ionize in water to produce surface-active anions. Unlike petroleum-derived counterparts, they use amino acids as raw materials, ensuring greater environmental and human friendliness.
From their nomenclature, anionic AAS are typically structured as:
Fatty acid + acyl + amino acid + potassium/calcium/sodium salt
The fatty acid component is provided by saturated and unsaturated fatty acids, commonly lauric acid, oleic acid, caprylic acid, palmitic acid, stearic acid, etc.
The amino acid part mainly includes sarcosine, glutamate, serine, alanine, leucine, valine, etc., with sarcosine and glutamate being the most used. Various combinations of fatty acids and amino acids yield diverse anionic amino acid surfactants.
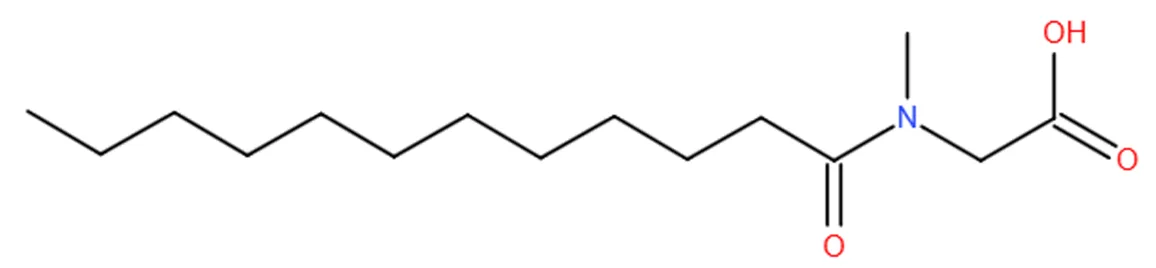
Synthesis Processes
Common synthesis processes for amino acids include three types: chemical synthesis, enzymatic synthesis, and chemo-enzymatic synthesis. Chemical synthesis boasts the largest capacity, subdivided into fatty acid chloride-amino acid reaction, fatty acid anhydride-amino acid reaction, fatty nitrile hydrolysis, etc. Enzymatic synthesis prepares N-acyl amino acids via enzyme catalysis, using lipases and proteases. E.L. Soo et al. used Lipozyme to catalyze lysine acylation with fatty acids, synthesizing N-ε-palmitoyl lysine and N-ε-oleoyl lysine, but yields remained around 30%, with high costs and difficult enzyme recovery unresolved. Thus, chemical synthesis dominates current production. Below is an introduction to chemical synthesis methods, updated with recent advancements as of 2025, including improved catalysts and greener processes for enhanced efficiency and sustainability.
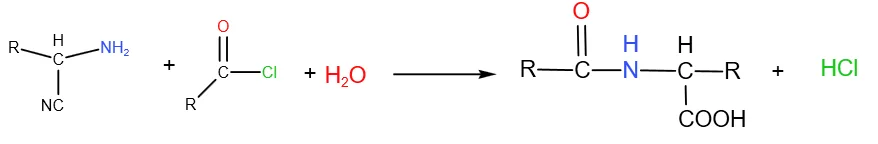
Fatty Acid Chloride and Amino Acid Reaction Process
N-acyl amino acids produced from fatty acid chlorides reacting with amino acid amino groups are the main industrial products. This is also known as the Schotten-Baumann condensation reaction, where fatty acid chloride and amino acid condense in alkaline aqueous solution or organic solvent to form N-acyl amino acid salt. Neutralization with inorganic acid, separation yields crude N-acyl amino acid; further alkaline neutralization gives purer product. The typical Schotten-Baumann reaction is as follows:
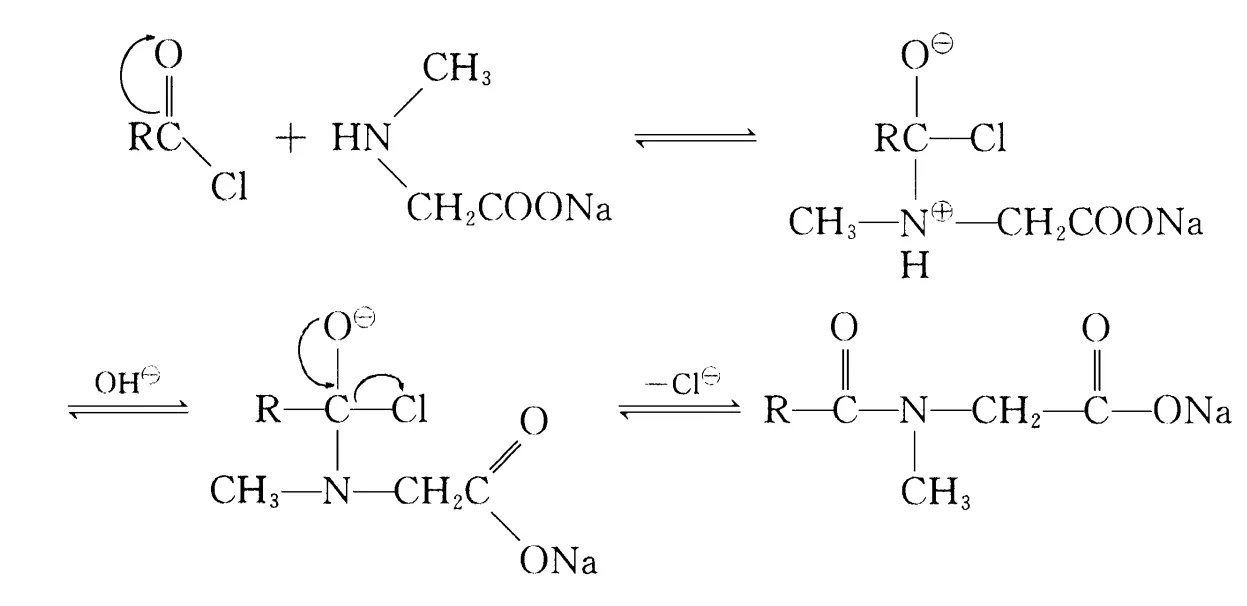
In this SN2 substitution, two side reactions may occur:
Fatty acid chloride hydrolysis:
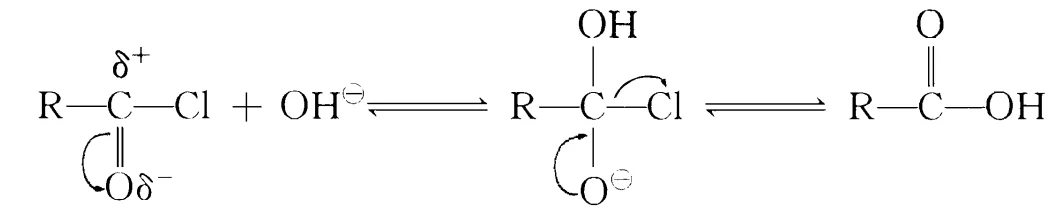
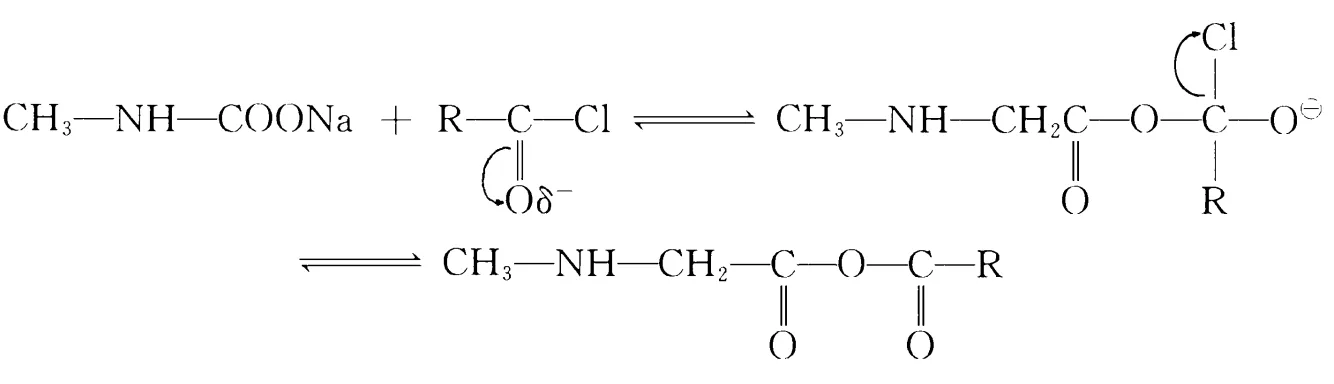
This requires high-quality acid chloride; hydrolysis produces higher fatty acids hard to remove, affecting quality. Thus, minimizing hydrolysis is key. Controlling pH during reaction and adding chlorides (one or more) reduces hydrolysis rate, minimizing side reactions. Xu Hujun et al. controlled OH– concentration by simultaneous dripping of acid chloride and sodium hydroxide; excess phosphorus trichloride in lauroyl chloride preparation increased yields.
Hassanzadeh et al. added organic solvents tetrahydrofuran and acetone, dripping acid chloride and sodium hydroxide for reaction at water-organic interface, reducing hydrolysis and late-stage viscosity. Reports indicate using tetrahydrofuran as solvent, 20-30°C, pH 10-11, achieves 85% conversion.
Recent 2025 advancements include cold-process methods to improve cost-efficiency and sustainability, as seen in ionic amino acid surfactant developments.
Fatty Acid Anhydride and Amino Acid Reaction Process
Yoshida R et al. in a patent proposed reacting anhydride and amino acid in stirred aqueous solution at certain temperatures (patent: -20 to 40°C) without catalyst to form N-acyl amino acid salt in water. Though simple, ensuring purity requires excess fatty acid anhydride, complicating separation; thus, no large-scale production yet.
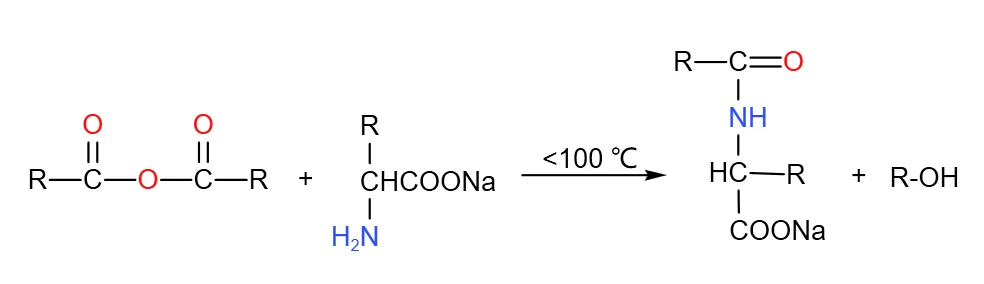
Fatty Nitrile Hydrolysis Acylation Process
This involves reacting fatty nitrile with fatty acid chloride via acid-binding agent in solvent to obtain N-acyl amino acid. Fatty nitrile is prepared via Strecker method, reacting aldehyde or amine with NaCN or HCN; acid chloride from fatty acid and chloride. High efficiency, yields over 95%, but complex process, extreme NaCN/HCN toxicity, HCN storage difficulties require on-site production near raw materials—preventing large-scale industrialization.
Amide Carbonylation Process
Since the 1970s, amide carbonylation for N-acyl amino acid salts has been researched in two steps: (1) Under acidic conditions, formaldehyde reacts with fatty amide to synthesize fatty acyl methylamino alcohol; (2) Under special catalyst (typically transition metal) and CO environment, alcohol reacts to amino acid salt.
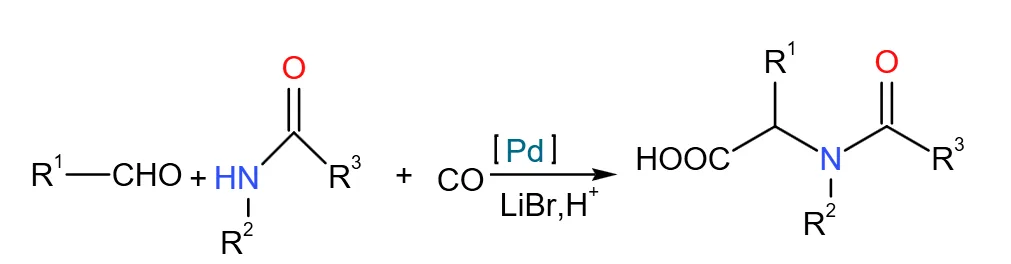
This uses readily available raw materials, good atom economy, high reaction rates. Matthias Beller et al. developed a palladium catalyst achieving 98% purity. Main issue: expensive, hard-to-recover catalysts contaminating products; developing new catalysts is key. Ryo Akiyama et al. used platinum catalysis for high yields, with no Pd residue detected in characterization.
Though not yet industrialized at scale, ongoing novel catalyst research may lead to future adoption. As of 2025, perspectives on aminoacylases in biocatalytic synthesis highlight enzymatic potentials for greener N-acyl amino acid surfactants.
For more details on surfactants, visit Surfactants. If you have questions, contact us. In summary, the synthesis processes of anionic amino acid surfactants evolve toward sustainability, with chemical methods dominating and enzymatic innovations promising eco-friendly advancements in anionic amino acid surfactants synthesis.
Summary of Surfactant Knowledge Points
A Series for Easy Understanding
Green surfactants
- Classification and Properties of Green Surfactants
- Natural Polysaccharide-Based Green Surfactants
- Properties and Applications of Anionic Amino Acid Surfactants
- Synthesis Processes of Anionic Amino Acid Surfactants
- Microbial Surfactants: Sophorolipids
- Types and Structures of Cardanol-Based Surfactants
New surfactants
- Synthesis and Applications of Phosphate Ester Surfactants
- Gemini Surfactants and Their Antibacterial Properties
- Low-Foam Surfactants: Types, Properties, and Applications
- Impact of Polyether Structure on Product Performance
- Types and Applications of Isomeric Alcohol Special Surfactants
- Research Progress on Secondary Alcohol Ethoxylates

