Cardanol, extracted from natural cashew nut shell liquid (CNSL) via advanced refining, is processed into various grades differing mainly in color, odor, and purity: refined CNSL, primary refined cardanol, and premium refined cardanol.
Cardanol features a unique chemical structure with these advantages:
- Benzene ring for high-temperature resistance;
- Polar hydroxyl group for wetting and activation on contact surfaces;
- Meta-position C15 straight chain with unsaturated double bonds for excellent toughness, hydrophobicity, low permeability, and self-drying properties.
- It can substitute or partially replace phenol in epoxy curing agents, phenolic laminates, copper-clad boards, and surfactants.
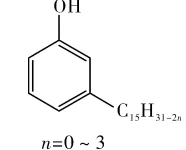
Cardanol’s structure (Figure 1) combines aromatic and aliphatic characteristics, while the phenolic hydroxyl imparts phenolic properties.
Cardanol-based surfactants offer superior biodegradability, known as green surfactants or bio-based surfactants, poised to replace petroleum-derived phenolics. Recent years have seen extensive research by domestic and international scholars, yielding promising results using cardanol as the lipophilic group.
Recent 2024-2025 studies highlight cardanol’s role in sustainable surfactants, including anion-nonionic hybrids and zwitterionics for enhanced performance in oil recovery and personal care.
Anionic Surfactants
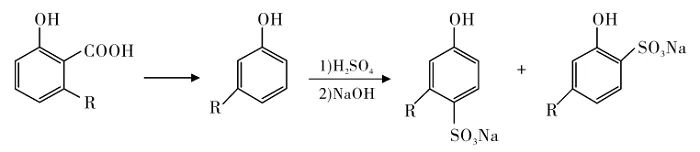
Cardanol Sulfonates
Peungjitton et al. synthesized cardanol sulfonate surfactants from CNSL, following the route in Figure 2.
Cardanol Sulfates
Bruce et al. reacted cardanol in methanol with vinyl sulfate to produce cardanol polyoxyethylene ether sulfate salt at 78% yield (Figure 3).
Cardanol Carboxylates
Scorzza et al. prepared cardanol carboxylate surfactants from cardanol (Figure 4). Sodium cardanol carboxylate yielded 41.5%, showing strong hydrophilicity, excellent surface activity, and low critical micelle concentration (CMC).
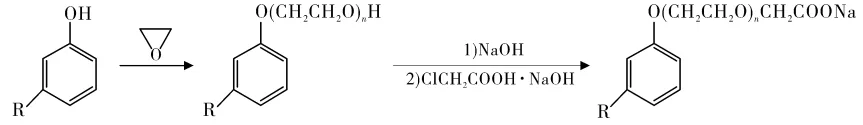
Cationic Surfactants
Wang Jun et al. used cardanol, epichlorohydrin, and trimethylamine to synthesize a novel cardanol cationic surfactant at 56% yield (80% quaternary ammonium content; Figure 5). Its CMC and γCMC were 4.83 mmol/L and 28.62 mN/m, demonstrating strong surface activity.
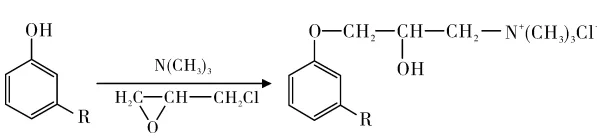
Nonionic Surfactants
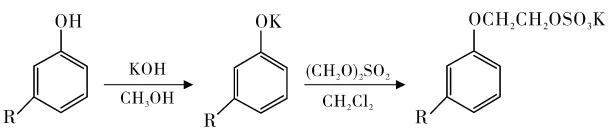
Cardanol Polyoxyethylene Ethers
Cardanol polyoxyethylene ethers represent a new generation of mild, safe, green surfactants—environmentally friendly nonionics. They synergize with anionics for enhanced wetting, penetration, emulsification, dispersion, and detergency. Rapid biodegradation suits detergents and personal care products.
Studies on blends with fatty alcohol polyoxyethylene ether (JFC) showed optimal metal cleaning at 2:3 ratio.
Tyman et al. reacted cardanol with ethylene oxide (Figure 6). Ethylene oxide adducts of 13-14 maximized surface tension reduction and biodegradability.
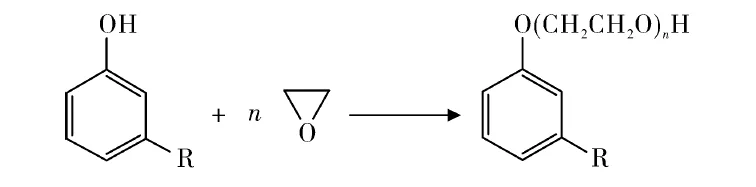
Unsaturated (UMPO) and saturated (SMPO) cardanol polyoxyethylene ethers of varying degrees fully replaced TX-10 as pesticide emulsifiers.
Cardanol-Based Glucosamides
A three-step reaction from cardanol yielded green nonionic cardanol-based glucosamide (Figure 7).
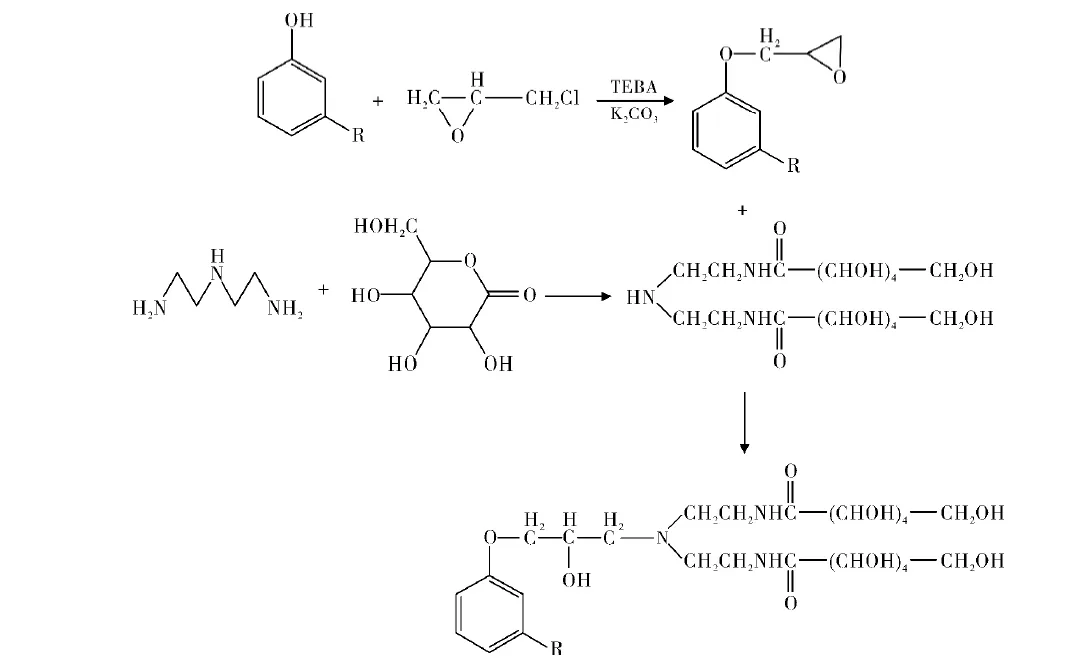
Trisiloxane-Containing Cardanol Ethers
Chloropropyl trisiloxane reacted with cardanol polyoxyethylene ether, followed by purification, to produce trisiloxane-containing cardanol ether surfactants (Figure 8).
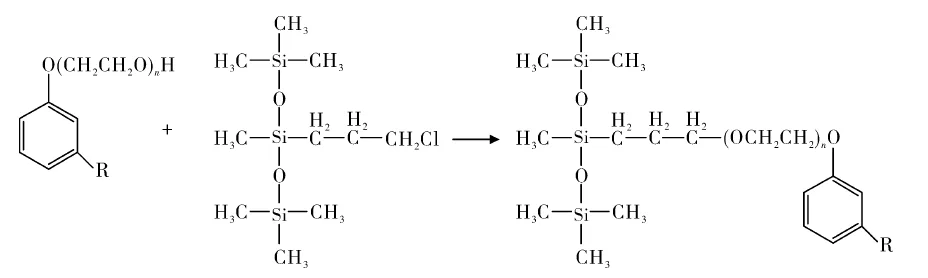
These combine silicone and cardanol properties: good solubility, low skin irritation, excellent biodegradability. Varying ethylene oxide adducts suit emulsifiers, wetting agents, solubilizers, and detergents.
Amphoteric Surfactants
Cardanol Chloroalcohol Ethers
Cardanol and epichlorohydrin, catalyzed by tetrabutylammonium bromide, formed intermediate cardanol chloroalcohol ether. Amination with dimethylamine yielded cardanol tertiary amine, then reaction with sodium 3-chloro-2-hydroxypropanesulfonate produced cardanol betaine amphoteric surfactant.
Tests showed CMC of 3.17 × 10-4 mol/L and γCMC of 32.86 mN/m.
At 0.5-3.0 g/L, it reduced oil/water interfacial tension to 10-4-10-3 mN/m. With polyacrylamide, tension dropped to 10-6-10-4 mN/m. Binary systems extended demulsification time to 200-1200 s (vs. 36 s alone), offering excellent emulsification for cost-effective oil displacement and residual oil recovery.
Applications and Development Prospects
As a natural long-chain phenolic, cardanol holds broad prospects in surfactants, attracting significant scholarly attention. Cardanol-based surfactants, as bio-based surfactants, excel in emulsification and detergency with low toxicity and biodegradability. Amid environmental protection and energy shortages, developing cardanol surfactants carries profound significance.
Recent 2025 research advances include trisiloxane hybrids and zwitterionics for enhanced sustainability in foaming, emulsification, and oil dispersion.
For more on related products, visit Surfactants. Questions? Contact us. In conclusion, cardanol-based surfactants drive innovation as green surfactants and bio-based surfactants, offering sustainable alternatives across industries.
Summary of Surfactant Knowledge Points
A Series for Easy Understanding
Green surfactants
- Classification and Properties of Green Surfactants
- Natural Polysaccharide-Based Green Surfactants
- Properties and Applications of Anionic Amino Acid Surfactants
- Synthesis Processes of Anionic Amino Acid Surfactants
- Microbial Surfactants: Sophorolipids
- Types and Structures of Cardanol-Based Surfactants
New surfactants
- Synthesis and Applications of Phosphate Ester Surfactants
- Gemini Surfactants and Their Antibacterial Properties
- Low-Foam Surfactants: Types, Properties, and Applications
- Impact of Polyether Structure on Product Performance
- Types and Applications of Isomeric Alcohol Special Surfactants
- Research Progress on Secondary Alcohol Ethoxylates