Gemini surfactants, also referred to as dimeric surfactants, twin surfactants, or coupled surfactants, are a novel class of surfactants formed by linking two traditional surfactant molecules at or near their hydrophilic head groups via a connecting linker. This unique structure endows gemini surfactants with exceptional properties not found in conventional surfactants, making them highly promising for diverse applications and a hot topic in colloid and interface science.
Structure of Gemini Surfactants
Gemini surfactants originated in the 1970s when Bunton first synthesized a series called methylene-α-ω-bis(dimethylalkylammonium bromide). Compared to traditional surfactants, gemini surfactants connect the ionic heads of two surfactant molecules to a linking group through chemical bonds. Structurally, they can be viewed as dimers of conventional surfactants, hence the term dimeric surfactants.
(Figure 1: Schematic diagram of surfactant structure)

Gemini surfactants exhibit higher surface activity than traditional ones, closely tied to their special structure. The presence of the linker increases the distance between charged head groups, reducing electrostatic repulsion while enhancing hydrophobic interactions among the tails.
This allows gemini surfactants to pack tightly at the air-liquid interface, reducing the adsorption area per molecule and preventing easy saturation, leading to greater adsorption and stronger micelle formation. However, with a rigid linker like a phenyl group, the interface curvature is smaller, and molecules align more loosely toward the air phase, weakening surface tension reduction compared to flexible linkers.
Solubilization Properties
Solubilization is a phenomenon linked to surfactant structure and micelle count, occurring only above the CMC. Structural differences affect solubilization efficacy. For gemini surfactants with identical hydrophobic chains, the solubilization order for hydrocarbons and polar organics is: nonionic > cationic > anionic.
This is mainly because nonionic types have lower CMC, and cationic ones may have looser groups than anionic. Studies show that nonionic gemini surfactants enhance solubilization of aliphatic hydrocarbons with longer hydrophobic chains but decrease it with longer hydrophilic chains, possibly related to aggregation numbers in solution.
Rheological Properties
The unique rheological behavior of gemini surfactants depends on their aggregate structures in aqueous solutions. At the same concentration, gemini surfactant solutions have much higher viscosity than traditional ones. Linker variations also influence rheology.
Short linkers yield peculiar rheology: viscosity surges with concentration (up to 6 orders of magnitude) before plummeting. This reflects micelle transitions from linear to network structures, then breakdown. Reiko’s research on symmetry shows that higher symmetry in gemini surfactants correlates with higher solution viscosity.
Types and Synthesis of Gemini Surfactants
Cationic Gemini Surfactants
Cationic gemini surfactants dissociate into cations in water, primarily ammonium or quaternary ammonium salts. They offer good biodegradability, strong detergency, chemical stability, low toxicity, simple structure, easy synthesis and purification, plus bactericidal, preservative, antistatic, and softening properties.
Quaternary ammonium gemini surfactants are typically prepared via alkylation of tertiary amines. Two main methods: (1) quaternization with dibromo-substituted alkanes and single long-chain alkyldimethyl tertiary amines; (2) using 1-bromo long-chain alkanes and N,N,N’,N’-tetramethylalkyl diamines in anhydrous ethanol under reflux. The second is preferred due to lower cost of dibromo compounds.

Anionic Gemini Surfactants
Anionic gemini surfactants dissociate into anions, including sulfonates, sulfate esters, carboxylates, and phosphate esters. They excel in detergency, foaming, dispersion, emulsification, and wetting, widely used as detergents, foaming agents, wetting agents, emulsifiers, and dispersants.
Sulfonates
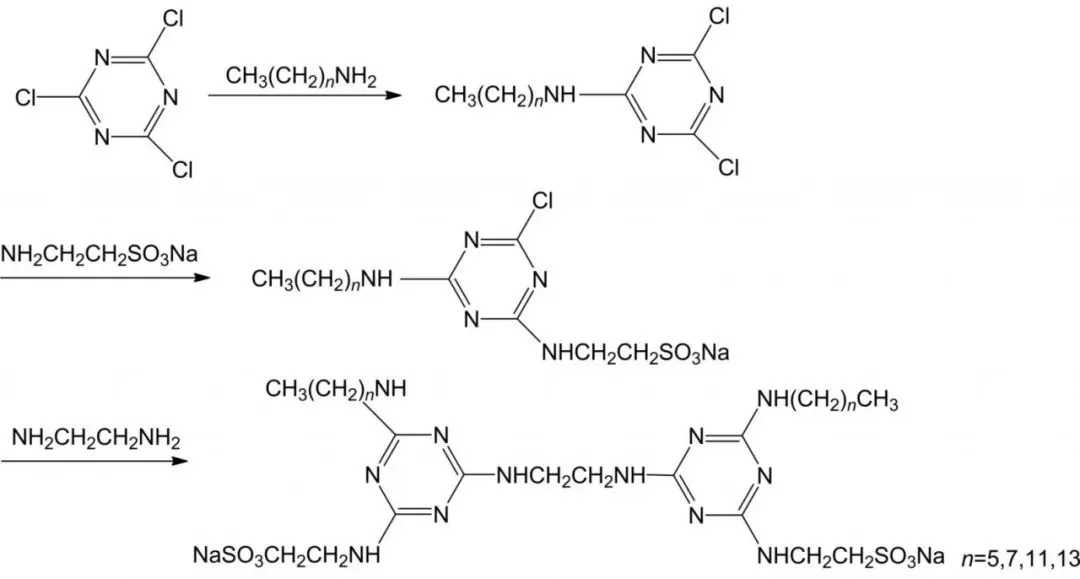
Sulfonate gemini surfactants boast good water solubility, wetting, heat and salt resistance, detergency, and dispersion, with broad raw material sources, simple production, and low cost. Applied in petroleum, textiles, and daily chemicals as detergents, foaming agents, etc. For example, Li et al. synthesized novel dialkyl disulfonic gemini surfactants (2Cn-SCT) from cyanuric chloride, fatty amines, and taurine via a three-step reaction.
Sulfate Esters
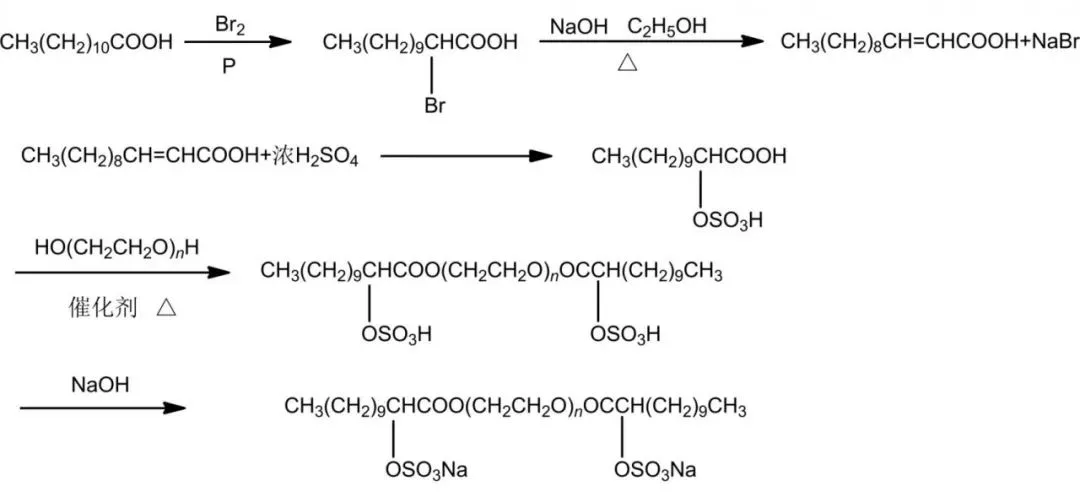
Sulfate ester gemini surfactants feature ultra-low surface tension, high activity, good solubility, broad sources, and simple synthesis. They offer excellent washing and foaming in hard water, neutral or slightly alkaline in solution. Sun et al. synthesized GA12-S-12 from lauric acid, polyethylene glycol, etc., via substitution, esterification, and addition to incorporate sulfate bonds.
Carboxylates
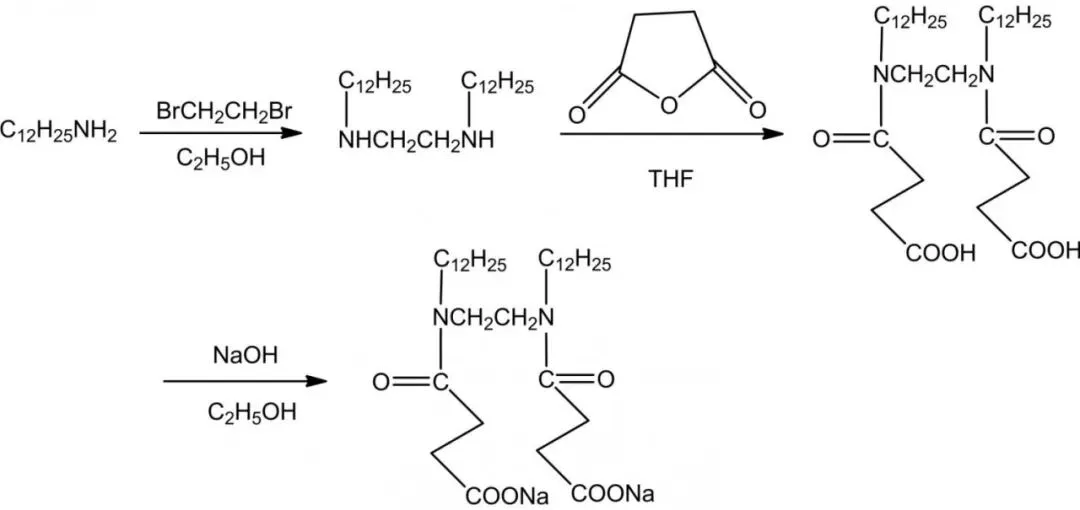
Carboxylate gemini surfactants are mild, eco-friendly, biodegradable, with natural sources, high metal chelation, hard water resistance, and calcium soap dispersion. They provide good foaming and wetting, used in medicine, textiles, and fine chemicals. Introducing amide groups enhances biodegradability, wetting, emulsification, dispersion, and detergency. Mei et al. synthesized CGS-2 with amide from dodecylamine, dibromoethane, and succinic anhydride.
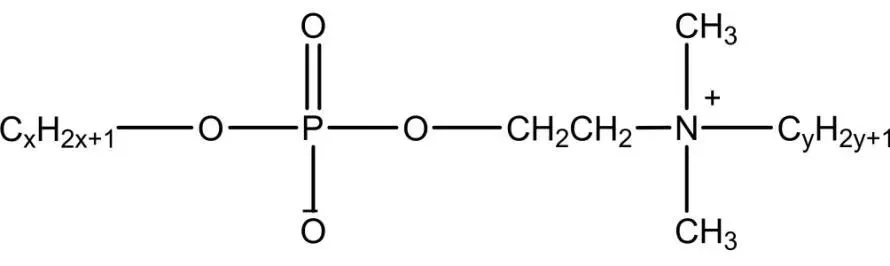
Phosphate Esters
Phosphate ester gemini surfactants mimic natural phospholipids, forming reverse micelles and vesicles. Used as antistatics, in laundry powders, with high emulsification and low irritation for skin care. Some have anticancer, antitumor, antibacterial properties, with dozens of drugs developed. Strong in pesticide emulsification for antibacterials, insecticides, and herbicides. Zheng et al. synthesized from P2O5 and n-octyl oligoglycol, offering good wetting, antistatic properties, simple process, and mild conditions.
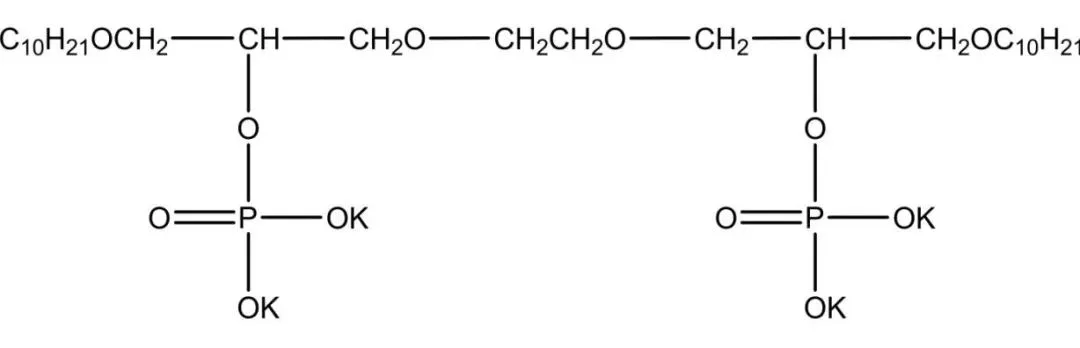
Nonionic Gemini Surfactants
Nonionic gemini surfactants do not ionize in water, existing molecularly. Research is limited, mainly sugar derivatives or alcohol/phenol ethers. They are highly stable, unaffected by strong electrolytes, compatible with other surfactants, and soluble.
Thus, they provide excellent detergency, dispersion, emulsification, foaming, wetting, antistatic, and bactericidal properties, used in pesticides, coatings, etc. In 2004, FitzGerald et al. synthesized polyoxyethylene gemini surfactants (GemnEm): (Cn-2H2n-3CHCH2O(CH2CH2O)mH)2(CH2)6.
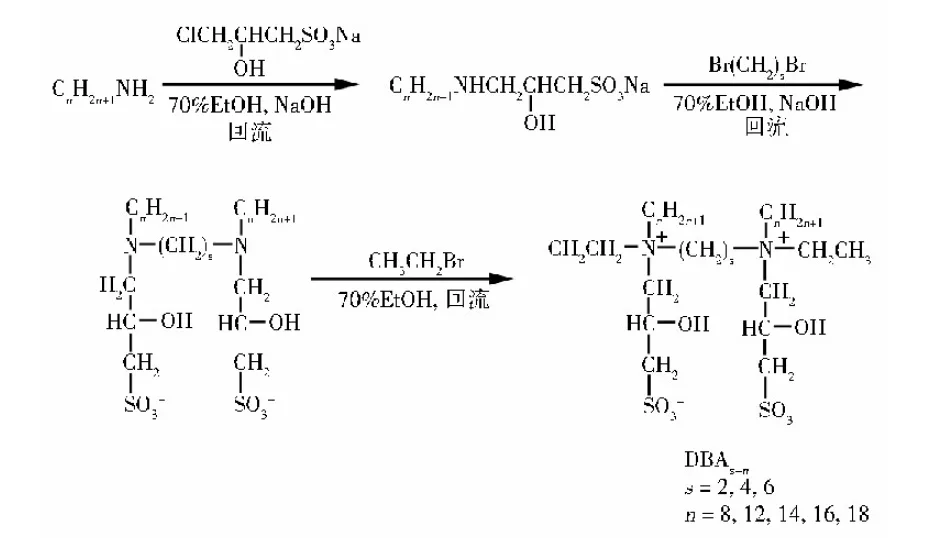
Zwitterionic Gemini Surfactants
Reported zwitterionics are mostly betaine or combined anionic-cationic types. Their dual charge centers enable better synergy with other gemini surfactants.
Geng et al. synthesized novel dihydroxy sulfate betaine (DBAs-n) from primary amines, 1,2-dibromoethane, etc., where linker length affects yield.
Applications of Gemini Surfactants
As interest in gemini surfactants grows, their unique efficiency expands applications: emulsifiers and dispersants in detergents, cosmetics and hygiene products, paint formulations, new material preparation, pharmaceuticals and biomedicine, enhanced oil recovery, flotation collectors, pollutant treatment (heavy metals, toxins), and soil remediation.
Detergents
Modern detergents rely on surfactants, and gemini surfactants excel in dissolving dirt with superior foaming, dispersion, and emulsification. Manufacturers favor them for detergency, processability, low cost, and eco-friendliness, though high production costs limit market share.
Personal Care Products
The structure of gemini surfactants enables interface enrichment, tension reduction, and property alteration. They encapsulate water-soluble ingredients for moisture retention, keeping skin hydrated, soft, and smooth. In cosmetics, they emulsify, disperse, solubilize, foam, and cleanse, meeting demands for specialized products.
Preparation of New Materials
With more hydrophobic groups, gemini surfactants self-assemble easily into diverse aggregates. They serve as templates and anti-adhesives for nanomaterials, outperforming in nanowires, nanotubes, and snowflake nanoparticles.
Metal Corrosion Protection and Inhibition
The amphiphilic structure forms interfacial films, aiding mechanical corrosion protection and batteries. Gemini surfactants alter metal oxide crystals, inhibit hydrogen evolution, delay electrode passivation, with concentration affecting performance.
For more details on surfactants, visit Surfactants. If you have questions, contact us. In summary, gemini surfactants continue to drive innovation across industries due to their superior performance and versatility.
Summary of Surfactant Knowledge Points
A Series for Easy Understanding
Green surfactants
- Classification and Properties of Green Surfactants
- Natural Polysaccharide-Based Green Surfactants
- Properties and Applications of Anionic Amino Acid Surfactants
- Synthesis Processes of Anionic Amino Acid Surfactants
- Microbial Surfactants: Sophorolipids
- Types and Structures of Cardanol-Based Surfactants
New surfactants
- Synthesis and Applications of Phosphate Ester Surfactants
- Gemini Surfactants and Their Antibacterial Properties
- Low-Foam Surfactants: Types, Properties, and Applications
- Impact of Polyether Structure on Product Performance
- Types and Applications of Isomeric Alcohol Special Surfactants
- Research Progress on Secondary Alcohol Ethoxylates

