Secondary alcohol ethoxylates, also known as secondary alcohol polyethers, are nonionic surfactants produced by ethoxylating secondary alcohols (typically C12-C15). These compounds stand out among nonionic surfactants for their low pour point, superior penetration, strong detergency/emulsification, and effective defoaming—making them highly versatile and eco-friendly alternatives to restricted alkylphenol ethoxylates (e.g., nonylphenol ethoxylates) and limitations of primary fatty alcohol ethoxylates or fatty acid ethoxylates.
Introduction to Secondary Alcohol Ethoxylates
Nonionic surfactants include alkylphenol ethoxylates, primary fatty alcohol ethoxylates, and fatty acid ethoxylates. However, alkylphenol ethoxylates (especially nonylphenol) face restrictions due to poor biodegradability and environmental toxicity. Primary fatty alcohol ethoxylates have high melting points (solid at room temperature), inconvenient use, and weaker penetration/detergency/emulsification. Fatty acid ethoxylates hydrolyze in alkali, limiting applications.
Secondary alcohol ethoxylates address these drawbacks. Synthesized from secondary alcohols (C12-C15) with 3-40 EO adducts (e.g., Japanese Catalyst SOFTANOL: 21% C12, 55% C13, 24% C14 secondary alcohols). Advantages: low pour point, strong penetration, excellent defoaming, high detergency/emulsification—widely used in household detergents and as safe, eco-friendly industrial surfactants.
Research Progress on Secondary Alcohol Ethoxylates
Synthesis involves two steps: secondary alcohol production, then ethoxylation.
Progress in Secondary Alcohol Synthesis
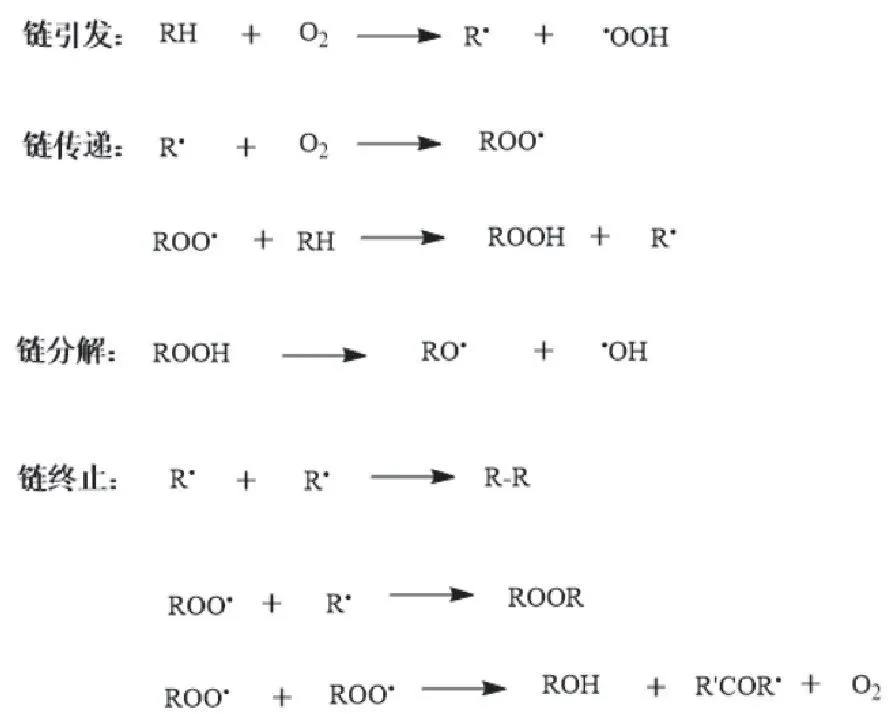
Liquid Wax Oxidation Method
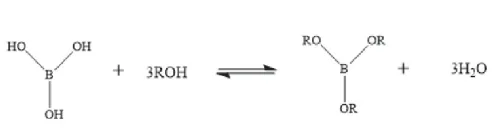
Influenced by boric acid crystal form: β-metaboric acid prevents catalyst aggregation/reduces tower blocking; γ-metaboric acid high melting point causes plugging—control <5%.
Batch or continuous oxidation viable.
Mid-1980s China: batch n-alkane oxidation trials → continuous studies determining equipment, flow, conditions, boric acid recovery. Per ton secondary alcohol: 1.599 t raw wax, 0.029 t boric acid, 0.107 t caustic.
Institutions examined temperature/time/boric acid/air oxygen/flow effects. Han Fei et al.: optimal 170-180°C, 4 h, 6% boric acid, 21% oxygen, 20-40 mL/min flow—~15% alkane conversion, ~83% secondary alcohol selectivity.
Olefin Addition Method
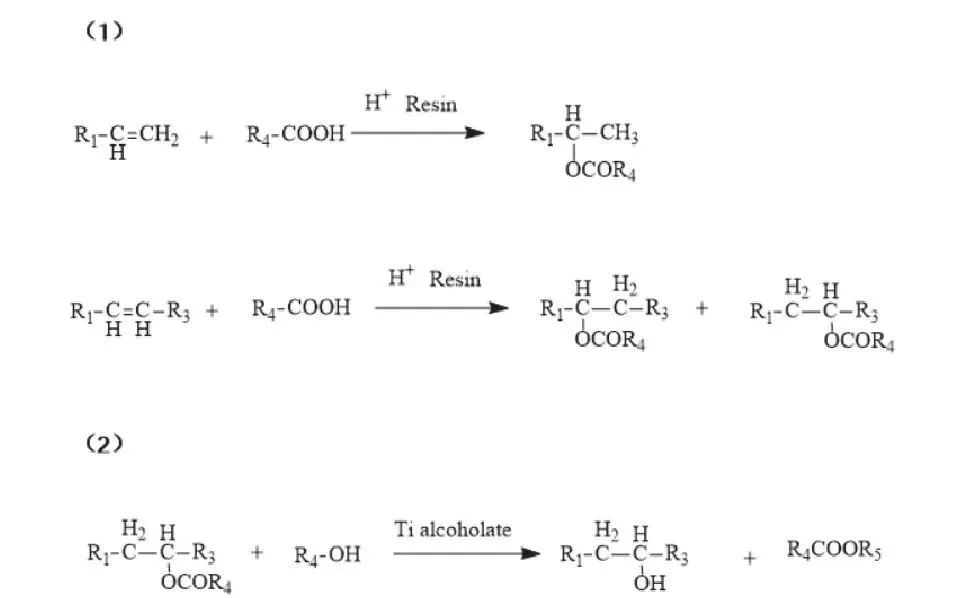
Union Carbide (US 4927954): acidic conditions carboxylic acid addition to α/internal olefins → ester; titanium chelate transesterification → secondary alcohol + new ester.
Isomerization yields varied structures + ester byproducts. Fixed-bed catalyst easy separation; continuous stable product. Lower impurities claimed.
Shell (CN102203049A) similar: acid-olefin → ester; special catalyst direct epoxide addition.
Steps: (1) internal olefin(s) + carboxylic acid(s) catalyst → ester(s); (2) ester + epoxide catalyst (oxygenated alkaline earth carboxylate/sulfuric/orthophosphoric acid/alcohol/ester) → acetylated secondary alkoxide; (3) hydrolysis/transesterification → secondary alkoxide.
Differs from UCC: epoxide addition post-ester, then optional hydrolysis/transesterification.
Dow: branched olefins → branched secondary alcohols; double metal cyanide catalyst → highly branched secondary alcohol polyethers: narrow MW distribution, low residual alcohol, low cost/waste—significant advance.
Aldol Condensation Method
CN201910566272.0: linear/tertiary-carbon-free branched aliphatic aldehydes self-condensation → branched enal; + tertiary-carbon-free aliphatic ketone organic base → branched dienone; hydrogenation → branched secondary alcohol.
Progress in Secondary Alcohol Ethoxylation
Conventional batch; continuous ethoxylation experimental—secondary alcohol only industrialized continuous.
Two-step: acidic catalyst (BF3) low EO (3-5) narrow distribution/low unreacted; alkaline (KOH) high EO (7/9/12).
Tubular reactors 2-stage, 4-5 atm, 50-70°C, 0.01-1% catalyst.
Dow (CN101918471A) similar: C12-15 secondary alcohols acidic low EO, alkaline high.
Carbon range: Japanese Catalyst SOFTANOL C12-14; Dow TERGITOL C12-15—broader tolerance, economic raw material flexibility. Japanese uses C12-14 n-alkanes oxidation—local availability.
1979 China interest; Light Industry Ministry Japan Catalyst exchanges.
Shanghai Light Industry Research Institute detailed: catalysts, oxidation, EO addition, recovery.
Rare earth catalysts reported (0.2% fluorinated rare earth).
CN201710252991.6: crude secondary alcohol alkali pretreatment + reactive distillation—simplifies, improves quality. Provides ethoxylation/refining.
Conventional: random EO addition Lewis catalyst low EO; remove acid/distill; alkaline further—complex.
Japanese Catalyst: linear olefins → secondary alcohol polyethers. Patent: polyalkylene glycol addition olefin double bond → higher secondary alkoxide; epoxide alkaline → epoxide adduct.
α-olefins zeolite/dodecylsulfonic acid isomerize internals (α 20-25%, internal 75-80%, ~15% single-pass). Skips acidic low EO—simpler.
Challenges secondary alcohol synthesis; Japanese Catalyst/Dow industrialized different routes. China 1980s deep basic research (universities/institutes/Yingkou pilot)—no commercialization then. Recently Jiangsu Saike sole domestic industrializer.
Outstanding performance drives attention; petrochemical advances abundant raw materials—increasing domestic focus, broader applications, more industrializers expected.
For more on related products, visit Surfactants. Questions? Contact us. In conclusion, secondary alcohol ethoxylates and secondary alcohol polyethers continue advancing as high-performance nonionic surfactants with expanding industrial potential.
Summary of Surfactant Knowledge Points
A Series for Easy Understanding
Green surfactants
- Classification and Properties of Green Surfactants
- Natural Polysaccharide-Based Green Surfactants
- Properties and Applications of Anionic Amino Acid Surfactants
- Synthesis Processes of Anionic Amino Acid Surfactants
- Microbial Surfactants: Sophorolipids
- Types and Structures of Cardanol-Based Surfactants
New surfactants
- Synthesis and Applications of Phosphate Ester Surfactants
- Gemini Surfactants and Their Antibacterial Properties
- Low-Foam Surfactants: Types, Properties, and Applications
- Impact of Polyether Structure on Product Performance
- Types and Applications of Isomeric Alcohol Special Surfactants
- Research Progress on Secondary Alcohol Ethoxylates

