The polyether structure plays a critical role in determining the performance of polyether-based products, including nonionic surfactants widely used in detergents, emulsifiers, defoamers, and lubricants. Factors such as initiator type, EO/PO ratio, polymerization mode (block vs. random), molecular weight, and end-capping directly affect hydrophilicity, hydrophobicity, viscosity, cloud point, and specialty functions. Optimizing polyether structure enables tailored solutions for industries like textiles, cosmetics, pharmaceuticals, and industrial cleaning.
Definition and Classification of Polyethers
Polyethers are high-molecular-weight polymers formed by ring-opening polymerization of epoxides with active hydrogen-containing compounds (initiators). Common epoxides include ethylene oxide (EO), propylene oxide (PO), butylene oxide (BO), and tetrahydrofuran (THF).
Depending on monomer type and conditions, polymerization yields:
- Block polyethers: EO and PO form distinct homopolymer segments arranged alternately (EPE or PEP types).
- Random polyethers: EO and PO distribute randomly along the chain, without long single-monomer segments.
Applications of Polyethers
Polyether nonionic surfactants are vital, offering excellent surface activity and adjustable HLB values. Used as lubricants, detergents, emulsifiers, dispersants, and defoamers in food, pharmaceuticals, cosmetics, fibers, and related industries.
Block Polyether Applications
EO/PO block polyethers are high-molecular-weight surfactants. Polyoxyethylene segments hydrophilic; polyoxypropylene (methyl-bearing) hydrophobic. Properties vary with molecular weight and EO/PO ratio—highly designable.
Recent uses: solubilization, extraction, biochemical separation, drug delivery, tissue engineering, mesoporous materials, pharmaceuticals, and cosmetics. Key areas: surfactant compounding, functionalization, biochemical separation, interfacial adsorption.
Random Polyether Applications
In EO/PO random polyethers, polyoxyethylene provides water solubility; polyoxypropylene maintains liquidity at room temperature. Exhibit cloud point behavior (phase separation on heating, resolubilization on cooling).
Applications: brake fluids, quenching agents, release agents, defoamers, lubricants for textiles, compressors, rubber processing. Widely in high-speed spinning oils: low heater residue, minimal white powder high-speed twisting, excellent wear resistance, low viscosity temperature dependence—main formulation component.
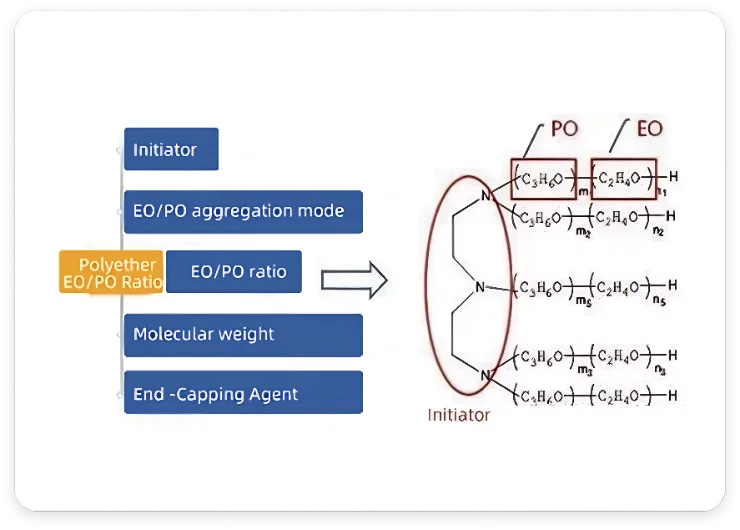
Relationship Between Polyether Structure and Performance
Polyether structure comprises five core elements: initiator, EO/PO ratio, polymerization mode, molecular weight, and end-capping agent.
Initiator
Polyethers form via epoxide ring-opening addition to initiators under catalysis. Initiator crucially impacts functionality and application.
Classified by functionality:
- Monohydric alcohols: n-butanol, isooctanol, lauryl alcohol, isotridecanol, stearyl alcohol → monofunctional polyethers (MW typically ≤2000). Used in high-speed spinning oils (e.g., butanol/C8-C18/C11-C13 isotridecyl polyethers). Allyl alcohol polyethers modify silicone fluids.
- Diols: propylene/ethylene glycol → difunctional polyethers, block types as high-molecular surfactants: emulsifiers, detergents, demulsifiers.
- Polyols/amines: glycerol, trimethylolpropane, pentaerythritol, sorbitol, sucrose; ethylenediamine, diethylenetriamine, triethanolamine → multifunctional for detergents, demulsifiers, polyurethane synthesis.
EO/PO Ratio
Significantly affects properties: higher EO stronger hydrophilicity; higher PO stronger lipophilicity.
For defoamers, EO typically ≤15%. Ratio designed per needs—tailors HLB/solubility/foaming.
EO/PO Polymerization Mode
Determines block polyethers vs. random polyethers.
Key differences:
- Penetration: Block far superior.
- Cloud Point: Block markedly lower—ordered arrangement stronger intermolecular forces, easier escape water “pull.”
- Thermal Weight Loss (210°C): Block higher—methyl steric hindrance reduces stability.
- Viscosity: Block significantly higher—ordered forces increase viscosity (consistent cloud point effect).
Block extremes yield qualitative performance leaps. E.g., penetration: random EO-end poor, PO-end improved—but block EO-end vastly superior all random (including PO-end).
Leverage for specialty additives: penetrants, defoamers in production.
Molecular Weight
Closely tied initiator functionality; limits vary. Monohydric typically ≤2000; multifunctional higher for foams/rigid applications.
End-Capping Agent
Beyond EO/PO capping, specialty end-capping (long-chain fatty acids, butanol) alters viscosity/surface tension for targeted functions.
For more on related products, visit Surfactants. Questions? Contact us. In summary, optimizing polyether structure—initiator, EO/PO ratio, mode, molecular weight, end-capping—directly enhances performance in diverse applications, driving innovation in polyether-based solutions.
Summary of Surfactant Knowledge Points
A Series for Easy Understanding
Green surfactants
- Classification and Properties of Green Surfactants
- Natural Polysaccharide-Based Green Surfactants
- Properties and Applications of Anionic Amino Acid Surfactants
- Synthesis Processes of Anionic Amino Acid Surfactants
- Microbial Surfactants: Sophorolipids
- Types and Structures of Cardanol-Based Surfactants
New surfactants
- Synthesis and Applications of Phosphate Ester Surfactants
- Gemini Surfactants and Their Antibacterial Properties
- Low-Foam Surfactants: Types, Properties, and Applications
- Impact of Polyether Structure on Product Performance
- Types and Applications of Isomeric Alcohol Special Surfactants
- Research Progress on Secondary Alcohol Ethoxylates

