Gemini surfactants, also known as dimeric surfactants, are a novel class of compounds featuring two amphiphilic moieties connected by a spacer near the head groups. Renowned for superior surface activity and unique antibacterial properties, gemini surfactants outperform traditional surfactants in efficiency and versatility. Their dual hydrophobic tails and hydrophilic heads enable tighter interfacial packing, lower critical micelle concentrations (CMC), and potent antimicrobial effects, making them ideal for sterilization, personal care, food production, and industrial applications.
Introduction to Gemini Surfactants
In 1991, Menger and Littau first synthesized rigid-spacer bis-alkyl chain surfactants, naming them Gemini surfactants. In China, Zhao Guoxi translated this as gemini surfactants, a term widely adopted. That year, Zana et al. prepared quaternary ammonium gemini surfactants and systematically studied their properties. By 1996, researchers summarized interfacial behavior, aggregation, rheology, and compounding with traditional surfactants. In 2002, Zana explored spacer effects on aqueous aggregation, significantly advancing the field. Qiu et al. later developed novel syntheses using cetyl bromide and 4-amino-3,5-dihydroxymethyl-1,2,4-triazole.
China’s research began later; in 1999, Zhao Jianxi’s review sparked interest, leading to rapid progress. Recent focus includes novel gemini surfactants, physicochemical properties, and applications in sterilization, food, defoaming, drug delivery, and cleaning.
Classified by head group charge: cationic, anionic, nonionic, and zwitterionic gemini surfactants. Cationic often quaternary/tertiary ammonium; anionic sulfonate/phosphate/carboxylate; nonionic polyoxyethylene; zwitterionic betaine/imidazoline.
Cationic Gemini Surfactants
Cationic gemini surfactants dissociate cations in water, mainly ammonium/quaternary types. They offer biodegradability, strong detergency, stability, low toxicity, simple structure/easy synthesis/purification, plus bactericidal, preservative, antistatic, and softening properties.
Quaternary gemini surfactants from tertiary amine alkylation: (1) dibromoalkane + long-chain alkyldimethyl tertiary amine; (2) 1-bromo long-chain alkane + N,N,N’,N’-tetramethylalkyl diamine in anhydrous ethanol under reflux. Second method preferred due to cost (Figure 2).

Anionic Gemini Surfactants
Anionic gemini surfactants dissociate anions: sulfonates, sulfates, carboxylates, phosphates. Excellent detergency, foaming, dispersion, emulsification, wetting—used as detergents, foaming/wetting/emulsifying/dispersing agents.
Sulfonates
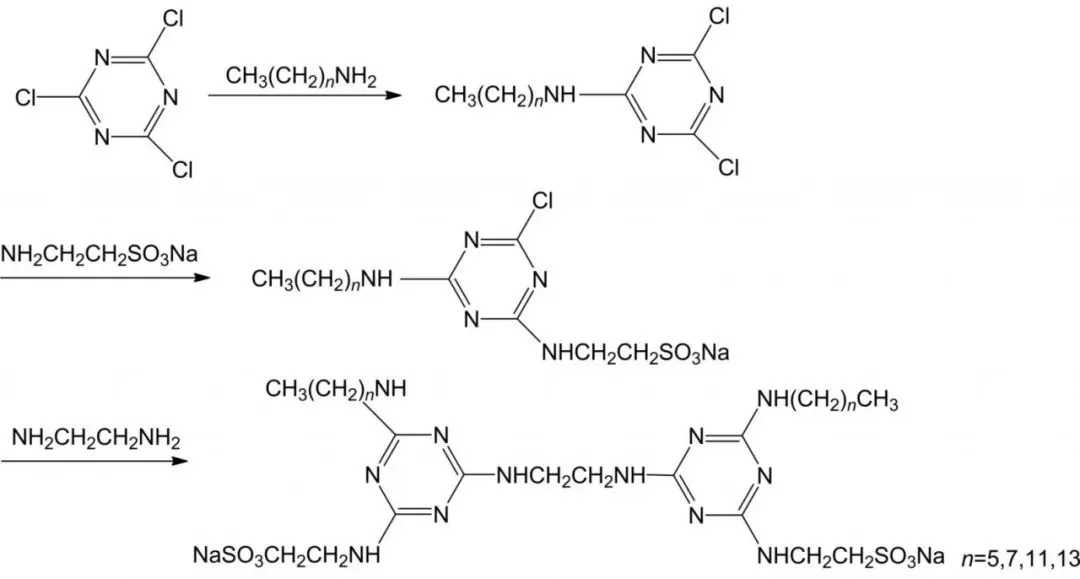
Sulfonate gemini surfactants: good solubility/wetting, temperature/salt resistance, detergency/dispersion; broad raw materials, simple/low-cost production. Widely in petroleum, textiles, daily chemicals (Figure 3). Li et al. three-step synthesis dialkyl disulfonic gemini surfactants (2Cn-SCT) from cyanuric chloride, fatty amines, taurine.
Sulfates
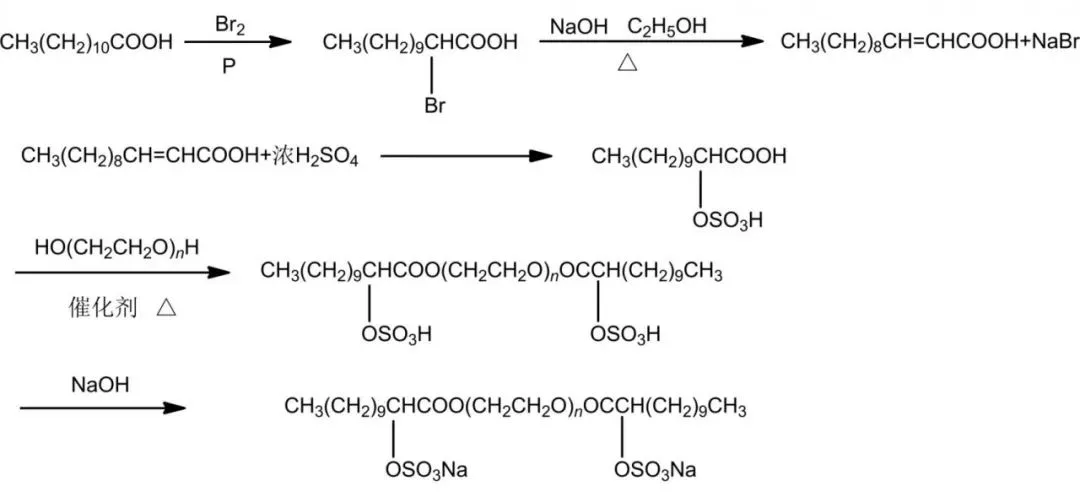
Sulfate gemini surfactants: ultra-low tension/high activity, solubility, broad sources/simple synthesis; good washing/foaming stable hard water, neutral/slightly alkaline (Figure 4). Sun et al. GA12-S-12 from lauric acid/polyethylene glycol via substitution/esterification/addition.
Carboxylates
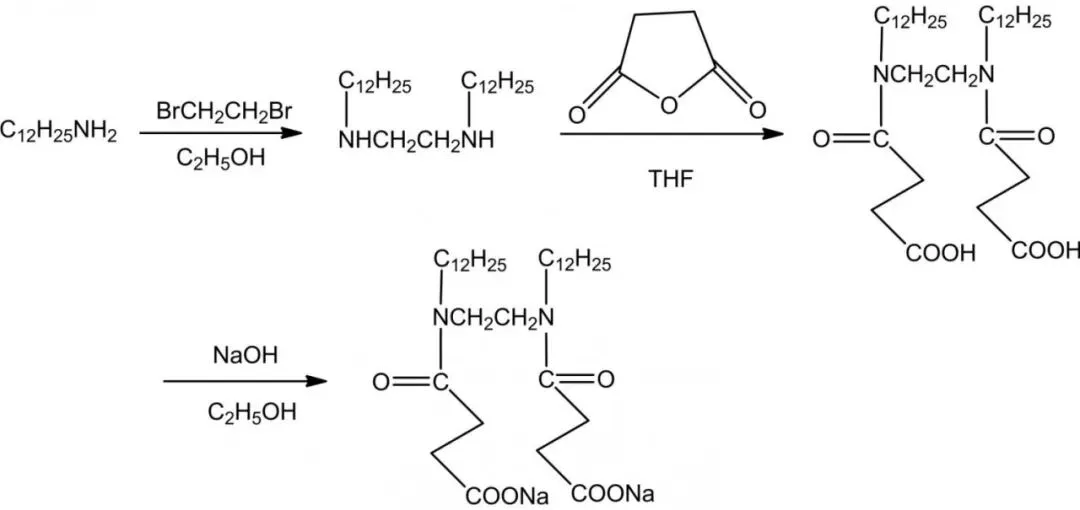
Carboxylate gemini surfactants: mild, eco-friendly, biodegradable natural sources; high metal chelation, hard water/calcium soap dispersion resistance; good foaming/wetting in medicine/textiles/fine chemicals. Amide groups enhance biodegradability/wetting/emulsification/dispersion/detergency (Figure 5). Mei et al. CGS-2 with amide from dodecylamine/dibromoethane/succinic anhydride.
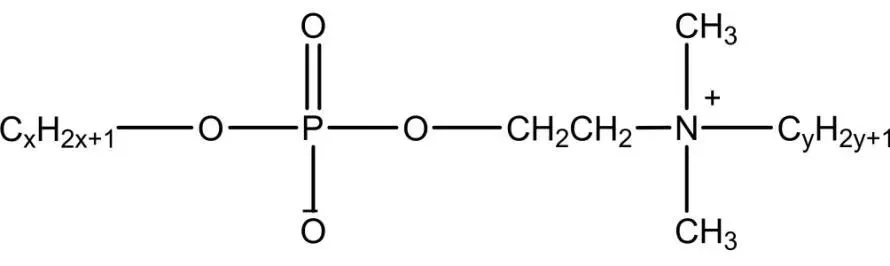
Phosphates
Phosphate gemini surfactants mimic phospholipids, forming reverse micelles/vesicles. Antistatics/laundry powders; high emulsification/low irritation skin care. Some anticancer/antitumor/antibacterial—dozens drugs. Strong pesticide emulsification antibacterial/insecticidal/herbicidal (Figure 6). Zheng et al. from P2O5/n-octyl oligoglycol: good wetting/antistatic, simple/mild process.
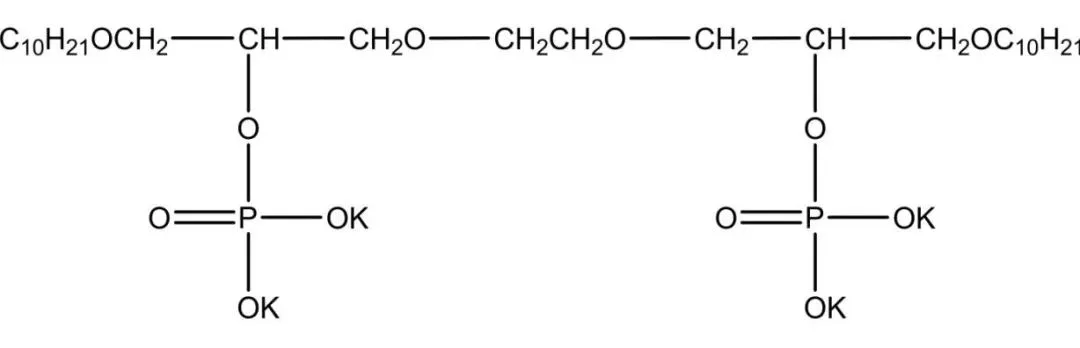
Nonionic Gemini Surfactants
Nonionic gemini surfactants non-dissociating molecular form. Limited research: sugar derivatives or alcohol/phenol ethers. High stability unaffected electrolytes, compatible, soluble.
Excellent detergency/dispersion/emulsification/foaming/wetting/antistatic/bactericidal—pesticides/coatings (Figure 7). FitzGerald et al. (2004) polyoxyethylene gemini surfactants (GemnEm): (Cn-2H2n-3CHCH2O(CH2CH2O)mH)2(CH2)6.
Physicochemical Properties of Gemini Surfactants
Surface Activity
Measured by aqueous surface tension reduction. Gemini surfactants pack directionally at interfaces (Figure 1(c)). CMC 2+ orders lower than traditional; C20 reduced. Dual heads maintain solubility long chains. Spacer covalent bond shortens head distance vs. traditional loose packing/repulsion—stronger activity.
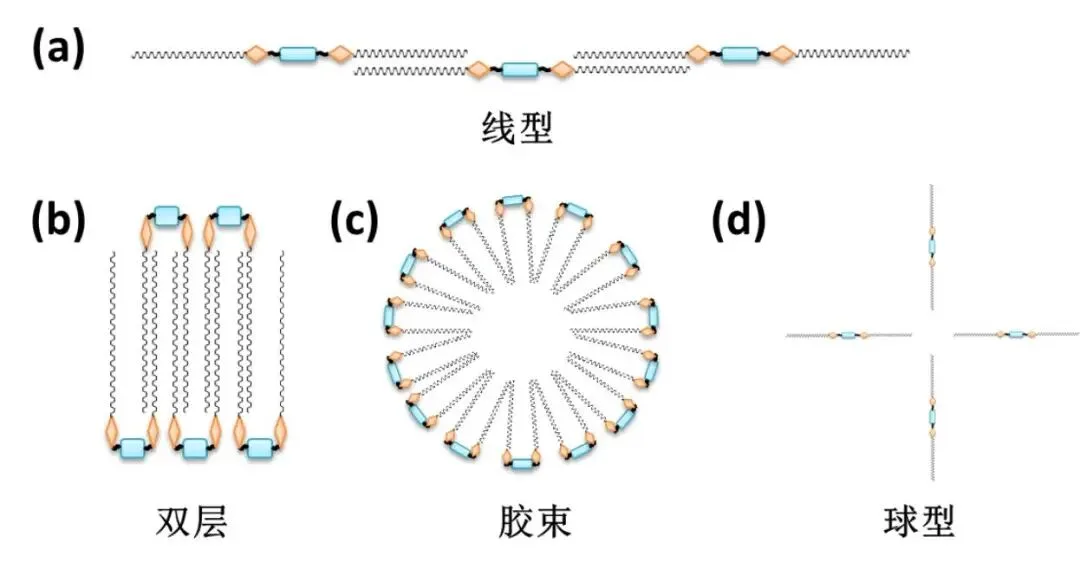
Assembly Structures
Above CMC, saturated surface forces internal migration forming micelles. Gemini surfactants form diverse morphologies: linear, bilayer vs. traditional spherical (Figure 9). Size/shape/hydration affect phase/rheology/viscoelasticity.
Traditional (e.g., SDS) spherical minimal viscosity impact. Gemini surfactants complex morphologies distinct solutions. Viscosity rises concentration—linear micelles entangle networks. Further increase drops—network breakdown other structures.
Antibacterial Properties of Gemini Surfactants
As organic antimicrobials, gemini surfactants bind microbial membrane anions or react thiols, disrupting proteins/membranes inhibiting/killing microbes.
Anionic Gemini Surfactants Antibacterial Properties
Inhibition from antibacterial groups. In latex/paints, hydrophilic chains bind water dispersants, hydrophobic hydrophobic dispersoids—dense protective film inhibits growth.
Mechanism differs cationic: solution/system/group-dependent, limited. Requires sufficient concentration ubiquitous killing; lacks targeting wastes, long use resistance.
Alkyl sulfonate gemini surfactants clinical: busulfan/treosulfan treat myeloproliferative—crosslink guanine/uracil unrepaired apoptosis.
Cationic Gemini Surfactants Antibacterial Properties
Mainly quaternary ammonium gemini surfactants. Strong bactericidal: dual hydrophobic chains adsorb cell wall (peptidoglycan); dual positive nitrogens promote adsorption negative bacteria, penetrating lipid layer altering permeability rupture; heads denature enzymes/proteins. Combined potent.
Environmentally: hemolytic/cytotoxic prolonged aquatic exposure, biodegradation increases toxicity.
Nonionic Gemini Surfactants Antibacterial Properties
Sugar derivatives or alcohol/phenol ethers.
Sugar: affinity binds membranes rich phospholipids. Sufficient concentration alters permeability pores/channels disrupting transport/exchange leakage death.
Phenol/alcohol ethers: target wall/membrane/enzymes halting metabolism/regeneration. Diphenyl ethers inhibit nucleic acid/protein enzymes limiting growth; paralyze metabolism/respiration exhaustion.
Zwitterionic Gemini Surfactants Antibacterial Properties
Simultaneous cationic/anionic, medium-dependent properties. Mechanism unclear—likely quaternary-like adsorption negative bacteria interfering metabolism.
Amino Acid-Type
Cationic zwitterionic dual amino acids—similar quaternary. Positive electrostatic attraction negative microbes; hydrophobics lipid bilayer leakage lysis death. Advantages: biodegradable low hemolytic/toxicity—expanding applications.
Non-Amino Acid-Type
Irreversible positive/negative centers: betaine/imidazoline/amine oxide. Betaine insensitive salts active acid/alkaline—acid cationic mechanism, alkaline anionic. Excellent compounding.
Conclusion and Outlook
Gemini surfactants increasingly applied due special structures: sterilization, food, defoaming, drug delivery, cleaning. Growing environmental demands drive eco-friendly multifunctional development.
Future: novel special structures/functions especially antibacterial gemini surfactants/antiviral; compounding traditional/additives superior products; cheap accessible raw eco-friendly synthesis.
For more on surfactants, visit Surfactants. Questions? Contact us. In summary, gemini surfactants with potent antibacterial properties revolutionize industries as efficient dimeric surfactants.
Summary of Surfactant Knowledge Points
A Series for Easy Understanding
Green surfactants
- Classification and Properties of Green Surfactants
- Natural Polysaccharide-Based Green Surfactants
- Properties and Applications of Anionic Amino Acid Surfactants
- Synthesis Processes of Anionic Amino Acid Surfactants
- Microbial Surfactants: Sophorolipids
- Types and Structures of Cardanol-Based Surfactants
New surfactants
- Synthesis and Applications of Phosphate Ester Surfactants
- Gemini Surfactants and Their Antibacterial Properties
- Low-Foam Surfactants: Types, Properties, and Applications
- Impact of Polyether Structure on Product Performance
- Types and Applications of Isomeric Alcohol Special Surfactants
- Research Progress on Secondary Alcohol Ethoxylates
