Natural polysaccharide-based green surfactants possess amphiphilic molecular structures, enabling strong adsorption at phase interfaces and improving compatibility between solid-liquid and liquid-liquid phases. Moreover, polysaccharides’ unique physicochemical properties provide excellent rheological behavior and gel network formation in solutions, allowing effective interfacial adsorption at low concentrations. They are commonly used for emulsification, dispersion, thickening, and stabilization in multiphase systems. Chemical modifications can balance hydrophilicity-hydrophobicity and impart chemical activity, surface charge, and rheological properties, yielding superior green surfactants. This article primarily introduces polysaccharides’ applications in surfactants, including cellulose, starch, hemicellulose, chitosan, and natural gums. Starting from their unique physicochemical properties, structures, and modification features, it analyzes their impacts on emulsification, dispersion, demulsification, and solubilization. It reviews research progress and application potential of polysaccharides and derivatives in polysaccharide surfactants. Finally, it highlights non-covalent bonding as a promising enhancement method for polysaccharide surface activity.
Polysaccharides are natural macromolecules formed by more than 10 monosaccharide units linked via glycosidic bonds, either linear or branched, with molecular weights ranging from thousands to millions. Depending on type and source, polysaccharides vary greatly in monosaccharide composition, glycosidic bond types, and branching structures. Additionally, hydroxyl groups on polysaccharides are often substituted by acetyl, amino, or methyl groups, endowing unique physicochemical properties.
Polysaccharides are the most abundant natural macromolecules, widely present in higher and lower plants, animals, and microorganisms, serving essential functions like energy storage, structural support, growth regulation, and signal transduction—indispensable for life activities.
Surfactants are chemicals that significantly reduce solvent surface tension or interfacial tension (e.g., liquid-liquid, liquid-solid, liquid-gas interfaces), offering emulsification, dispersion, wetting, solubilization, and stabilization effects, widely applied in food, chemical, agricultural, and construction industries. Studies show polysaccharides’ unique saccharide structures provide good water-binding and certain amphiphilicity, forming stable adsorption layers at incompatible interfaces (liquid-liquid, solid-liquid), reducing interfacial tension. Polysaccharides’ abundant hydroxyl structures form hydrogen bonds with water or intermolecularly, increasing aqueous solution viscosity or forming interwoven networks, providing stable steric barriers for emulsion droplets or solid particles, reducing collisions and aggregation to maintain sol dispersion stability.
For macromolecular surfactants, higher molecular weight reduces surface activity, so polysaccharides typically exhibit low surface activity. Even recognized high-performance natural polysaccharides like gum arabic and pectin have limited amphiphilicity, leaving room for improving interfacial adsorption. To better utilize polysaccharides in surfactants, physicochemical modifications are needed to enhance water solubility, amphiphilicity, rheology, surface activity, and network formation in solutions.
This article selects the most common and abundant natural polysaccharides, including plant-derived cellulose, starch, hemicellulose; animal-derived chitosan; and widely used natural gum polysaccharides in surfactants. It reviews recent research progress on polysaccharides and modifications in surfactant applications, explaining their unique emulsification and dispersion capabilities through structural analysis.
Cellulose
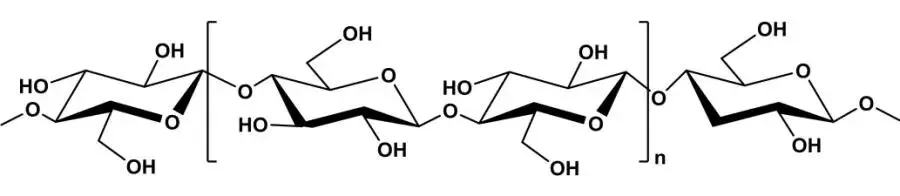
Cellulose, widely present in plant cell walls providing mechanical strength to stems, has a chemical structure as shown in Figure 1. Cellulose chains consist entirely of D-glucopyranose units linked by β-1,4 glycosidic bonds, with polymerization degrees from thousands to tens of thousands. Chains form hydrogen bonds via hydroxyl groups on structural units, creating crystalline fiber bundles. However, cellulose’s high crystallinity inhibits water solubility, making uniform stable systems difficult without structural disruption. Thus, conventional cellulose requires processing, mainly nanosizing fiber bundles or functionalizing surfaces to enhance water solubility and amphiphilicity for dispersing solid or oily particles in water.
Cellulose Micro-Nano Particles
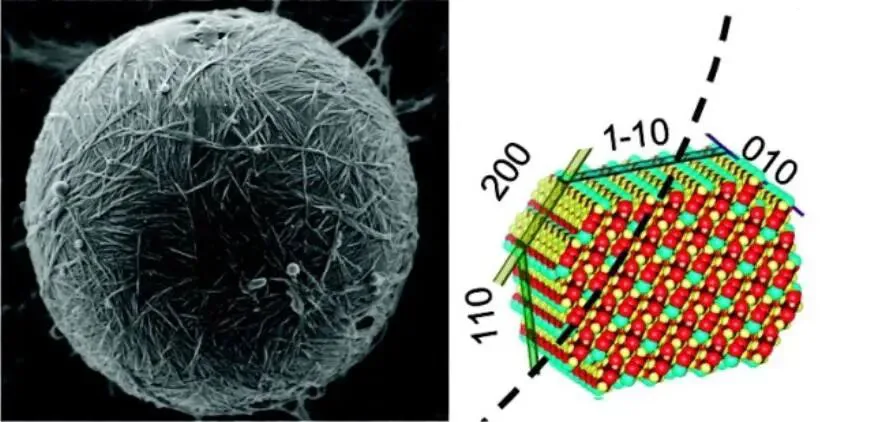
Hydrolyzing amorphous regions of cellulose fiber bundles via chemical or biological methods yields crystalline particles sized as cellulose nanocrystals (CNC) or microcrystals. These micro-nano crystals have hydrophobic surfaces and hydrophilic amorphous edges, conferring unique amphiphilicity as surfactants distinct from solid particle emulsifiers, forming stable Pickering emulsions (Figure 2). In formation, CNC crystalline regions adsorb well at interfaces with high adsorption energy, considered thermodynamically stable.
In emulsions, CNCs gain surface charge from carboxyl or sulfonic groups via acid hydrolysis, causing electrostatic repulsion between wrapped particles or droplets—a key stability factor. However, controlling CNC surface charge shows excessive density (>0.03 e/nm²) hinders oil/water interface stability. Thus, managing CNC surface charge density is crucial for emulsion stability. CNC morphology also affects stability during adsorption. Different crystal forms yield varying hydrophilicity-hydrophobicity, a key stability metric. Controlling preparation to adjust CNC geometry and crystal structure aids surface adsorption on oil droplets, vital for superior emulsification; as in Figure 2, the (200) plane is hydrophobic—if oriented at wedge-shaped CNC bases, better for spherical droplet adsorption. Introducing lower-crystallinity celluloses like cellulose nanofibrils (CNF) or regenerated cellulose increases emulsion viscosity, reducing droplet collisions and enhancing stability.
For solid-liquid dispersions, amphiphilic CNCs stably adsorb at interfaces, reducing tension. High steric hindrance prevents particle collisions, so CNCs disperse and stabilize high-surface-energy nanoparticles like carbon nanotubes, which aggregate easily in solution.
Modified Cellulose
Functionalizing cellulose disrupts natural crystallinity, improving water solubility, and introduces hydrophilic/hydrophobic or active groups to tune amphiphilicity, solubility, surface activity, and rheology for surfactant applications.
Hydrophilic Functionalization
Hydrophilic functionalization enhances water solubility while forming stronger amphiphilic structures with residual cellulose crystals. Increased solubility expands molecular gyration radius and hydroxyl-water interactions, boosting viscosity for stable networks via steric stabilization. It includes ionic (selective oxidation, carboxymethylation) and non-ionic (hydroxymethylation, hydroxyethylation, hydroxypropylation) groups.
Ionic hydrophilic celluloses like carboxymethyl cellulose (CMC) have high viscosity and surface charge, building macromolecular barrier networks and stabilizing dispersions via charge repulsion—a common macromolecular surfactant. CMC improves sand dispersion in cement, enhancing adhesion, used as stabilizers and hydrophilizing agents in cement and building materials. In oil-water emulsification, CMC minimizes droplet migration via viscosity increase, delaying collisions and coalescence. CMC binds oppositely charged agents for stable particle encapsulation; its thickening reduces gravity settling in double-layer encapsulations, enhancing stability—making CMC a good oil-water emulsifier.
Notably, excessive carboxymethyl substitution in CMC overly increases hydrophilicity, reducing amphiphilicity and viscosity. Polymerization degree variations affect viscosity and rheology, impacting emulsion stability. Balancing substitution and polymerization is key for surfactant applications.
Excessive hydrophilic functionalization reduces amphiphilicity and viscosity, hindering interfacial adsorption. In cases, hydrophobic groups are needed to tune molecular amphiphilicity and viscosity. Li et al. grafted long carbon chains on hydroxyethyl cellulose for hydrophobic modification, studying rheology and dispersion stability. Results showed high concentration-dependent viscosity and elevated apparent viscosity at low concentrations. Electrolytes and shear enhanced hydrophobic association within ranges, showing good thermal stability.
Hiranphinyophat et al. modified hydrophilic CNC with cyclopentane for high-surface-activity grafted polymers, exhibiting good thermoreversibility, repeating at least 3 emulsification/demulsification cycles without significant droplet loss or size increase.
Hydrophobic Functionalization
Esterification, etherification, and grafting introduce hydrophobic groups to cellulose hydroxyls. These interact hydrophobically, adsorbing cellulose at liquid-liquid/solid interfaces, enhancing surface activity and preventing aggregation via steric hindrance for stable sol dispersions.
Methylcellulose (MC), a key commercial cellulose ether, offers good water dispersibility, gelation, and surface activity, widely used as dispersants, emulsifiers, thickeners, and suspending agents in food, bio, and construction. MC surface activity depends on substitution degree and methyl distribution along chains, with low molecular weight dependence—but higher weights aid solid particle adsorption. Maintaining high molecular weight at high methyl substitution favors MC in surfactants. MC’s non-ionic nature stabilizes viscosity over wide pH, with good colloid/particle stability. Oil-water emulsification shows MC reduces interfacial tension, adsorbs stably with considerable thickness; high viscosity aids further stability. MC aggregation and network formation in water indicate temperature rise causes helical molecules to aggregate into linear fibrous aggregates, entangling into networks and multiplying viscosity.
Notably, during interfacial adsorption—especially oil-water—MC’s short hydrophobic chains fail to penetrate droplets internally, causing instability; longer chains are needed. Compared to MC, ethylcellulose (EC) has longer chains and better activity, often as water-in-oil demulsifiers. EC forms stable adsorptions on water droplets, with chains bridging multiple droplets, leading to flocculation and demulsification for oil-water separation.
Hydroxypropyl methylcellulose (HPMC), substituted by methyl and hydroxypropyl, resembles MC with similar rheology but hydroxypropyl provides higher associative steric hindrance, suppressing fibrous aggregate formation at higher temperatures for faster entanglement. HPMC’s longer hydrophobic segments penetrate oil droplets better, occupying larger areas for stronger adsorption and stability—indicating longer hydrophobic chains enhance polysaccharide surfactant emulsification.
Crosslinking cellulose derivatives further promotes macromolecular networks, better stabilizing sols via steric barriers. Huang et al. grafted saponified epoxidized soy protein onto hydroxyethyl cellulose for water-in-oil emulsions, finding grafted derivatives significantly reduce tension, with higher activity than CMC, achieving good stability at low concentrations. Crosslinking alters solubility, viscosity, and rheology; controlling degree is crucial.
Starch
Starch, one of Earth’s most utilizable and economical organics, shares composition with cellulose—D-glucose units—but linked by α-1,4 bonds versus cellulose’s β-1,4, causing vast physicochemical differences. Classified as branched (amylopectin) or linear (amylose), amylopectin dominates natural starch (75%-80%). Starch has higher water solubility than cellulose, especially amylopectin, dissolving at low temperatures and gelatinizing at high for viscosity surges—used as thickeners, emulsifiers, and binders in food and papermaking.
Natural Hydrocolloids
Starch gelatinization involves suspended particles absorbing water during heating, separating chains from crystalline states; upon completion and cooling, chains reassociate into 3D gel networks. Gelatinized starch acts as stabilizers for particle/droplet dispersion, positively correlating gelatinization degree with dispersion/emulsification; low degrees yield poor performance. Under shear, gelatinized starch shows low emulsion yield stress due to irreversible polymer network damage.
Beyond stabilization, helical starch hydroxyls orient outward, forming internal cylindrical hydrophobic cavities for hydrophobic binding with carbon nanotubes; outward hydroxyls reduce solid-liquid tension for stable dispersion. Starch enhances surface activity via non-covalent encapsulation in helices (Figure 3). De Fenyol et al. complexed vanillin with amylose helices via hydrogen bonds, stably adsorbing at interfaces with superior air-liquid tension reduction.
Modified Starch
Hydrophilic Functionalization
Hydrophilic functionalization yields etherified products like carboxymethyl starch, hydroxypropyl starch, and cationic starch, enhancing solubility and swelling for better gelatinization. Modified starch gelatinizes easier due to hydrophilicity; upon cooling, functional groups sterically prevent recrystallization for superior effects.
Sodium carboxymethyl starch (CMS), a common anionic starch ether, resembles CMC with superior performance, substitutable in industries as surfactants. Hydroxypropyl starch, non-ionic, uses propylene oxide for etherification, with low gelatinization temperature (usually 40°C) and good dispersion/thickening. As etherification products, CMS and hydroxypropyl starch properties/applications depend on substitution degree. Low degrees yield high apparent viscosity and gelatinization temperature; increases lower both.
Cationic starch, from tertiary/quaternary ammonium etherification, has similar gelatinization to CMS but viscosity rises with substitution, exceeding natural starch—benefiting emulsion stability.
Hydrophobic Functionalization
For effective activity, starch needs higher amphiphilicity; hydrophobic modifications enhance interfacial adsorption. Typically via esterification/grafting, hydrophobized starch shows strong polarity—especially dual hydrophilic-hydrophobic for high activity.
Alkenyl succinic anhydride esterification is widespread for starch hydrophobization. Hydrophobic groups weaken interchain hydrogen bonds; bulky groups reorganize structures via steric hindrance, increasing flexibility for water penetration/swelling, lowering gelatinization temperature/effects. Reports show alkaline-modified starch swelling rises with temperature/substitution due to weakened hydrogen bonds allowing more hydroxyl-water interactions. Enhanced swelling/water retention favors emulsifiers/colloid stabilizers. Chen et al. reacted hydrophobic groups on starch via hydrothermal, finding treatment prioritizes surfaces/amorphous regions with slightly higher peak viscosity than traditional. Studies show modified starch viscosity highly molecular weight-dependent, somewhat substitution-influenced; droplet size/stability mainly depend on molecular weight/average chain length. Balancing substitution/molecular weight is key for surfactant applications.
Recent advances confirm starch modifications like esterification enhance green surfactant applications in Pickering emulsions.
Chitosan
Chitin, nature’s second-most abundant macromolecule after cellulose, comprises N-acetylglucosamine linked by β-1,4 bonds, distributed in crustacean shells, insect cuticles, cephalopod endoskeletons, and fungal walls. Acetylated amino groups make chitin nearly water-insoluble; deacetylation enhances solubility. Deacetylated product is chitosan, classified by degree: low (55%-70%), medium (70%-85%), high (85%-95%), ultra-high (95%-100%), with solubility increasing accordingly. Chitosan has lower polymerization (hundreds to thousands) and abundant amino groups than cellulose, offering higher reactivity, solubility, and amphiphilicity. Yet, even highly deacetylated, chitosan dissolves poorly except in acid; excessive deacetylation reduces amphiphilicity—requiring hydrophilicity-hydrophobicity balance.
Natural Amphiphilicity of Chitosan
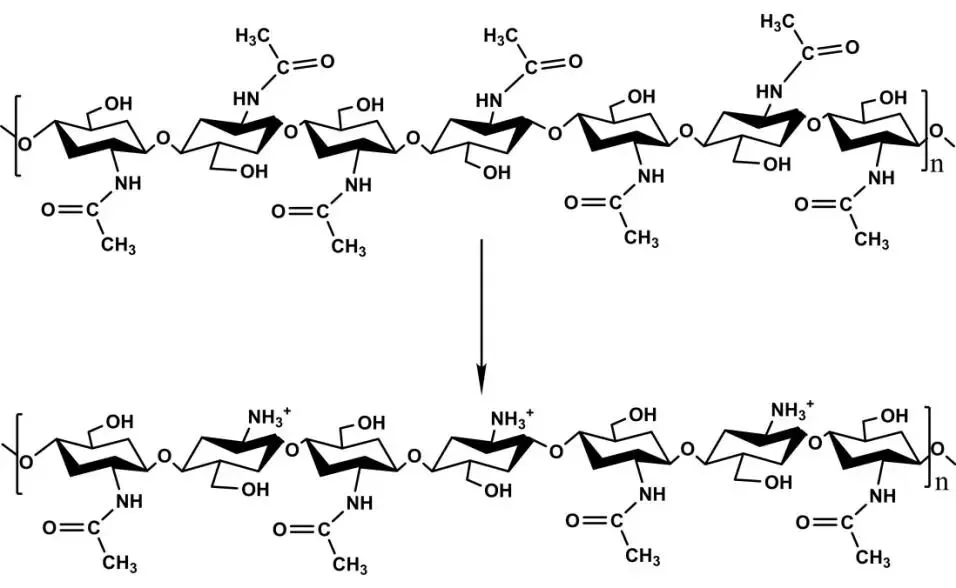
Chitosan’s abundant amino groups make it poorly soluble neutrally/alkalinely, but protonation in acid boosts hydrophilicity. Controlling deacetylation imparts hydrophobic groups; acid protonation increases solubility, combining for amphiphilicity (Figure 4), enhancing activity.
Unlike low pH, at higher pH (>pKa), amino deprotonation reduces net charge. Reduced interchain repulsion and strong acetyl attractions cause self-aggregation, enhancing hydrophobicity. Aggregation degree determines particle size/amphiphilicity—key for oil-water adsorption; it also tunes dispersion viscosity for emulsion stability. Self-aggregation suits chitosan as food stabilizers/gelling agents. Notably, amino deprotonation is reversible, enabling pH-induced demulsification.
Modified Chitosan
Even in acid, highly deacetylated chitosan has low dispersibility; chemical modifications enhance solubility and pH response range.
Carboxymethylation effectively boosts solubility/charge. Carboxymethylated chitosan dissolves well at high pH with good oil-water emulsification, but in acid, intramolecular/intermolecular electrostatic coupling reduces performance. Grafting quaternary ammonium increases solubility/charge. Hou et al. synthesized cationic modified chitosan emulsifier via ring-opening/quaternization, showing reduced tension and increased viscosity in asphalt emulsification.
Studies show long-chain comb copolymers better compatibilize interfaces than short diblocks; increasing “teeth” number/length reduces immiscible phase tension. Thus, long-chain hydrophobic modification replaces short acetyls to boost activity. It alters rheology, changing water-in-oil emulsion to weak gel characteristics.
Notably, electrostatically coupling protonated chitosan amines with oppositely charged polymers enhances amphiphilicity, reducing chemical pollution while boosting activity. Lasareva et al. coupled hexadecyl-oligo-oxyethylene hemisuccinate (HOS) with chitosan in acid, yielding HOS-chitosan with high emulsification/stability—due to stronger hydrophobic interactions/electrostatic repulsion forming stable anionic associations in compact spheres.
Overall, chitosan’s hydrophilic modifications consider anionic groups’ electrostatics; long-chain hydrophobics cause self-association but suit Pickering emulsifiers. Controlling chain length/amphiphilicity per needs is vital.
Recent studies confirm chitosan and hemicellulose as eco-friendly surfactants in pesticide delivery and environmental applications.
Hemicellulose
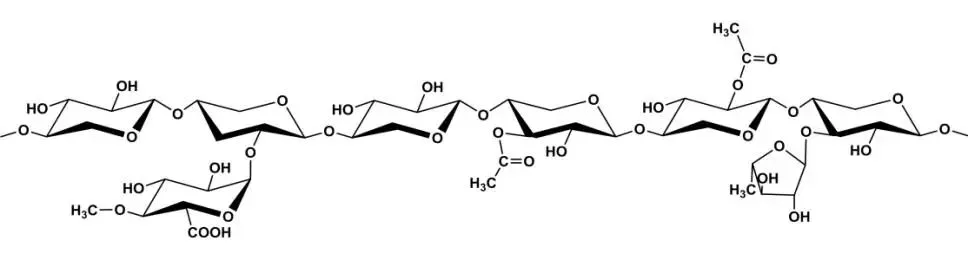
Hemicellulose, a cell wall polysaccharide class encompassing all heteropolysaccharides except cellulose, starch, and pectin, exists in nearly all plant cell walls—Earth’s second-most abundant natural macromolecule after cellulose, highly renewable. It fills/bridges cell wall components (cellulose, lignin, proteins) for specific structures and growth regulation. Classified by main chain monosaccharides/linkages into xylans and mannans, with possible galactose, arabinose, glucuronic acid branches. Xylans (GX) have β-D-xylopyranose main chains linked by β-1,4 bonds, branching degree/type varying by source (Figure 5); mannans (GGM) have β-D-glucopyranose/β-D-mannopyranose (ratios vary) linked by β-1,4, usually low-branching linear.
Natural Surface Activity
Compared to cellulose/starch/chitosan, hemicellulose has higher branching/solubility, high viscosity at low concentrations—excellent hydrocolloids. Natural hemicellulose has high acetylation, varying by extraction, providing hydrophobicity as macromolecular surfactants for particle/droplet dispersion/stabilization. GX/GGM significantly reduce emulsion oil-water tension, enhancing water-in-oil formation/stability. Compared to commercial stabilizers (Tween 20), GGM boosts viscosity for months-long stability. Lower molecular weight hemicellulose forms smaller uniform droplets, preventing coalescence. Especially GGM tends to intra/intermolecular assemblies, possibly forming Pickering stabilization—superior to GX.
Some cereal bran hemicelluloses covalently link proteins, forming amphiphilic xylan-protein gums as good emulsifiers/dispersants. Xiang et al. extracted amphiphilic arabinoxylan-protein gum (APG) from distiller’s grains, comparable to gum arabic in emulsification.
However, natural activity suffices limitedly (acetyls easily removed in alkali); appropriate chemical modifications needed. Modifications enhance amphiphilicity/activity, imparting stronger emulsification/dispersion—key hemicellulose application direction.
Modified Hemicellulose
Most hemicelluloses’ diverse structures yield varying solubility/activity; targeted enhancements broaden applications.
Hydrophilic Functionalization
Extraction removes substituents/branches, increasing chain regularity/partial crystallization—hindering dispersion. Hydrophilic modifications via etherification boost solubility. Carboxymethylation effectively enhances; improved dispersions don’t significantly reduce phase tension but surface charge forms strong electrostatic repulsion for solid particle dispersion. Hydroxypropylation also boosts solubility, reducing tension more evidently with higher substitution.
Hydrophobic Functionalization
Hydrophobic groups crucial for interfacial adsorption; low content yields low activity—needing hydrophobization for higher amphiphilicity. Yet, hemicellulose often has poor dispersibility; further hydrophobization worsens—requiring hydrophobics without solubility loss.
Succinic anhydride acylation provides both hydrophilic/hydrophobic groups, effectively boosting amphiphilicity. It adds charge/hydrophobicity for higher repulsion/activity; in oil-water emulsification, lower droplet sizes/higher stability. Emulsification correlates with substitution—excessive not better. Xiang et al. succinylated APG, yielding emulsifier rivaling gum arabic, with superior stability at pH>5—indicating high activity/emulsification potential.
Plant Gums
Natural plant gum polysaccharides are versatile hydrocolloids from exudates, seeds, seaweeds; often partially chemically bound to proteins/lipids for amphiphilicity—as thickeners, adhesives, emulsifiers, dispersants in food/daily chemicals.
Pectin
Pectin, an acidic hydrocolloid, widely in higher plant roots/stems/leaves/fruits’ primary/secondary walls, key for growth/development. Exact structure debated; main chain highly soluble D-galacturonic acid linked by α-1,4 bonds, interrupted by L-rhamnose causing discontinuities. Comprises 17 monosaccharides like D-galacturonic acid, D-galactose, L-arabinose via 20 linkages; main chain 2,6-positions O-acetyl substituted, 6-position methyl esterified for partial hydrophobicity—but limited impact; interfacial adsorption mainly from ferulic acid esters in branches/crosslinked proteins’ hydrophobicity. In emulsification, proteins/ferulic acids adsorb on droplets reducing tension; hydrophilic main chains wrap externally, stabilizing via steric/electrostatic repulsion (Figure 6).
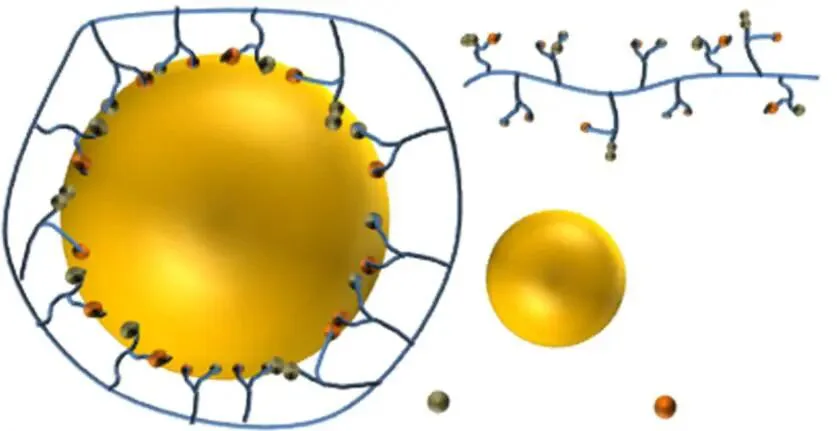
To confirm protein impact on pectin activity, regulate content/nature. Increasing protein sharply reduces droplet size, stabilizing—attributed to more adsorption reducing tension; excess saturates without further gain. Excess protein associates, hindering unfolding/adsorption at interfaces; connection structure affects interfacial behavior. Some argue protein presence doesn’t ensure good emulsification—accessibility/chemistry (amino acid composition/conformation) decisive.
Thus, increasing pectin protein boosts activity. Pectin’s uronic units provide negative charges; in acid, couple electrostatically with oppositely charged proteins (e.g., whey) for higher-protein complexes. Complexes have higher adsorption rates/lower viscosity than native, adsorbing faster/stably for smaller droplets.
Molecular weight/side chain content also affect emulsion viscosity, droplet coverage/stability. Funami et al. compared heat-treated pectin; enhanced side chains more stable, indicating treatment provides greater stability.
Past studies deemed acetyl irrelevant to emulsification; recent find at low protein, acetyl presence significantly improves. Main chain methyl also correlates with activity. Schmidt et al. proved direct relation between pectin methylation degree/emulsification. Notably, excessive methylation causes hydrophobic self-association, reducing performance—but milder than long chains, acceptable in cases.
Gum Arabic
Gum arabic, a plant exudate gum, 80% from Acacia senegal—oldest industrial gum. Structurally (Figure 7), neutral-slightly acidic complex polysaccharide; main chain β-D-galactopyranose linked by 1,3 bonds; side chains 3-5 β-D-galactopyranose via 1,3 linked to main via 1,6. Grafted hydrophobic proteins confer natural amphiphilicity; three main components: arabinogalactan (AG, 88% gum weight), arabinogalactan-protein (AGP, ~10%), glycoprotein (GP, <2%). AGP has high MW (~1500 kDa) and ~10% protein, strongest interfacial activity. As Figure 8, hydrophobic protein AGP adsorbs well on oil droplets, interweaving 2D networks preventing aggregation.
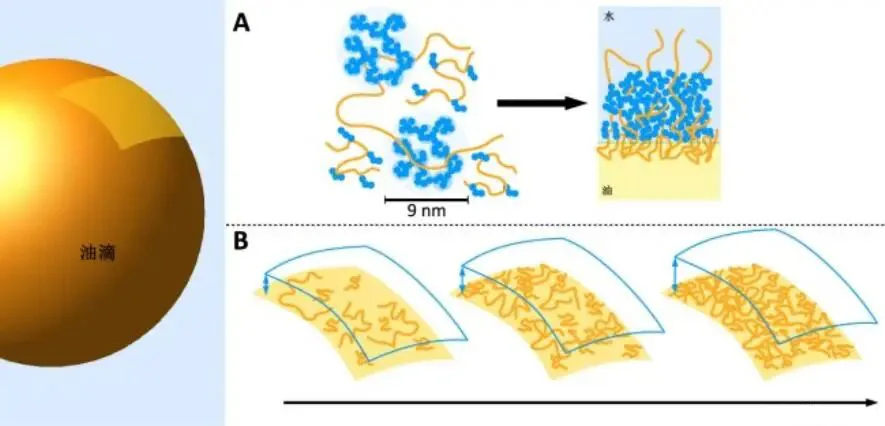
On emulsion droplet surfaces, polypeptide chains extend into oil, polysaccharide into water—forming thick hydrophilic coatings for strong steric anti-aggregation stability. Increasing gum arabic boosts interfacial adsorption, reducing/uniformizing droplet sizes. Beyond oil-water, APG activity disperses nano-solids uniformly/stably.
For further activity, chemical modifications. Shi et al. hydrophobized with octenyl succinic anhydride; modification increased MW, viscosity with substitution, greatly enhancing activity/stability.
Guar Gum
Guar gum, natural neutral polysaccharide from guar bean endosperm post-dehulling/degerming—a typical galactomannan. Structurally, main chain β-D-mannopyranose linked by β-1,4; branches α-D-galactopyranose via 1,6 to main. Highest MW natural water-soluble polysaccharide (1×106-2×106), high viscosity—even 1% dispersion reaches 10 Pa·s—used as industrial stabilizers, emulsifiers, thickeners.
Carboxymethylation etc. introduces more hydrophilic groups/charges, enhancing water-binding/electrostatic stabilization. Hydrophobics improve mortar rheology, increasing shear-thinning/consistency. Long-chain hydrophobized guar suits oil-water emulsification/particle dispersion. Hydrophobics reduce crystallinity, boosting dispersibility. Especially alkenyl succinic anhydride-modified guar has excellent amphiphilicity, thermal stability, thickening—potential gum arabic substitute.
Plant gums like pectin, gum arabic, guar gum advance sustainable surfactants in Pickering emulsions and eco-applications.
Summary and Outlook
Recently, to reduce fossil consumption/environmental impact, using renewable biodegradable polysaccharide surfactants as synthetic alternatives offers economic/environmental benefits. Polysaccharides are abundant/diverse; select per needs or modify chemically for superior products. But modifications inevitably introduce environmental/biological toxicity—especially in food, requiring caution. E.g., alkenyl succinic anhydride good for amphiphilic modification, but product biosafety needs verification.
Non-covalent bonding like electrostatic coupling/hydrophobic association instead of covalent chemical modifications improves amphiphilicity, yielding better polysaccharides and expanding reagent choices—e.g., safe proteins—greatly reducing impacts on biocompatibility/safety. Such bonding between polysaccharide types yields synergies for superior composites. Thus, non-covalent bonding holds great potential for enhancing polysaccharide surface activity.
For more on related products, visit Surfactants. Questions? Contact us. In conclusion, natural polysaccharide-based green surfactants like those from cellulose, starch, and gums drive innovation in sustainable industries, offering eco-friendly surfactants with minimal ecological footprint.
Summary of Surfactant Knowledge Points
A Series for Easy Understanding
Green surfactants
- Classification and Properties of Green Surfactants
- Natural Polysaccharide-Based Green Surfactants
- Properties and Applications of Anionic Amino Acid Surfactants
- Synthesis Processes of Anionic Amino Acid Surfactants
- Microbial Surfactants: Sophorolipids
- Types and Structures of Cardanol-Based Surfactants
New surfactants
- Synthesis and Applications of Phosphate Ester Surfactants
- Gemini Surfactants and Their Antibacterial Properties
- Low-Foam Surfactants: Types, Properties, and Applications
- Impact of Polyether Structure on Product Performance
- Types and Applications of Isomeric Alcohol Special Surfactants
- Research Progress on Secondary Alcohol Ethoxylates
