Sophorolipids are an important class of microbial surfactants that offer green, sustainable alternatives in various industries. Join us as we delve into the world of sophorolipids from BookChem.
Introduction to Sophorolipids
Research on sophorolipids began in the 1960s, slightly later than rhamnolipids. They were first isolated by Gorin et al. from the culture medium of T. magnolia as hydroxy fatty acid-containing sophorose glycosides. In 1968, this yeast was reidentified as T. apicola, also known as C. apicola. Subsequently, many teams have studied various strains of yeast or fungi capable of producing sophorolipids. Currently, European companies like Evonik and Croda have achieved industrial production of sophorolipids; Japan’s Saraya company applies sophorolipids in detergent products; and France’s Soliance incorporates sophorolipid components in cosmetics.
Sophorolipids are mixtures secreted by yeast, consisting of multiple sophorolipid molecules. Natural yeast-produced sophorolipids have over 20 major structures and more than 100 minor ones. Different structures and compositions of sophorolipids exhibit varying physicochemical activities, enabling their use in diverse fields.
Structure of Sophorolipids
The hydrophilic part of sophorolipids is the glycolipid moiety, while the hydrophobic part consists of saturated or unsaturated long-chain hydroxy fatty acids. Sophorolipids can be classified into different types based on the length, saturation, acetylation position, and degree of the fatty chains. A common classification is based on the presence of 1,4” esterification, dividing them into lactonic and acidic forms.

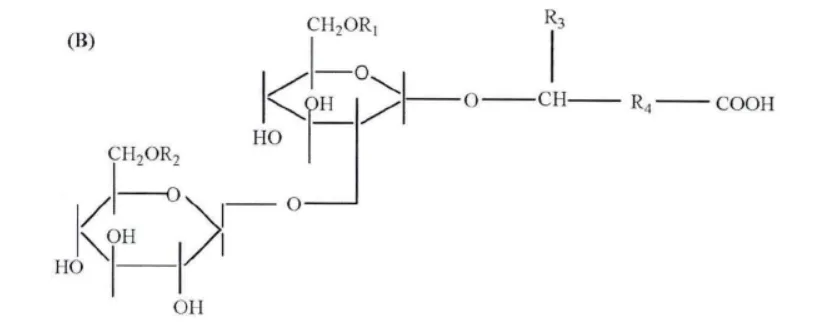
The sugar moiety of sophorolipids provides excellent hydrophilicity, while the fatty acid chain offers hydrophobicity. This combination gives sophorolipids strong surface activity, reducing water’s surface tension from 72.8 to 40 mN/m at a critical micelle concentration of 40-100 mg/L. The HLB value of sophorolipids ranges from 10-13, making them suitable as emulsifiers for oil-in-water emulsions.
Notably, current research on sophorolipids focuses mainly on antibacterial, anticancer, and spermicidal properties. Different structures of sophorolipids have unique performances: lactonic forms exhibit stronger antibacterial and anticancer activities, while acidic forms have superior antiviral capabilities. Additionally, as the degree of acetylation in sophorolipid molecules increases, their biological activity enhances accordingly.
Synthesis Pathways and Fermentation Processes
The synthesis of sophorolipids requires various non-pathogenic yeasts. Through screening and identification by researchers, strains such as Torulopsis magnolia (now Candida apicola), Torulopsis gropengiesseri, Torulopsis bombicola, and Candida bogoriensis have been identified. In recent years, students have also cultivated and screened new production strains.
The synthesis pathways and selected strains for sophorolipids vary depending on the carbon source. Common carbon sources include glucose and fatty compound sources, hydrocarbons as the sole source, and glucose as the sole source.

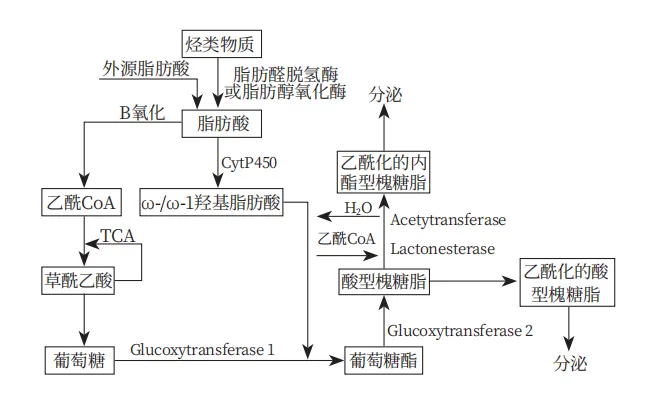

In addition to synthesis pathways, the choice of fermentation process is crucial. Fed-batch fermentation is the most common process for sophorolipid production. Compared to traditional batch fermentation, fed-batch significantly increases sophorolipid yields. Davila et al. found that supplying sufficient hydrophilic carbon sources and then feeding fatty acid esters could boost sophorolipid yields from 255 g/L to 340 g/L.
Semi-continuous fermentation is another commonly used process. When product levels are high, the product-rich fermentation broth is discharged to downstream separation, followed by adding fresh medium and reactants to the reactor. This method cycles repeatedly, but after certain batches, the yeast’s production capacity declines sharply. Researchers have used this method for 7 cycles, achieving a carbon source conversion rate of 0.74. This approach effectively reduces auxiliary time for batch changes, mitigates product inhibition to some extent, and alleviates foaming issues in fermentation. However, as fermentation broth is discharged, some yeast is lost, requiring time and materials for regrowth.
Yeast loss can extend production cycles and increase raw material costs. To minimize losses, researchers have developed continuous fermentation, which retains microorganisms in the reactor while separating products. Thus, efficient separation has become a research hotspot.
The above methods are all liquid fermentation techniques. In later stages, due to the foaming nature of the product, foaming is inevitable. Although semi-continuous and continuous methods reduce foam by separating products, more hydrophobic substrates (which eliminate foam) must still be added. Solid-state fermentation avoids foam generation. The Jiménez-Peñalver group used polyurethane foam as a substrate for solid-state fermentation, achieving good yields.
However, in practical production, large-scale solid-state fermentation reactor design requires more research. Notably, solid-state fermentation can use waste solid materials as raw materials or address environmental issues.
Separation Methods
The various fermentation methods mentioned above require separating sophorolipids afterward. Common separation methods include gravity separation, foam flotation, extraction, chromatography, recrystallization, and membrane filtration. Gravity separation relies on sophorolipids precipitating at the reactor bottom in later stages, with heating further increasing recovery rates, but the product purity is low, suitable for low-purity applications like oil recovery. Solvent extraction is the most common for biosurfactant separation; lab-scale uses ethyl acetate extraction, and after chemical pretreatment, single-structure products can be extracted. Crystallization targets acetylated lactonic sophorolipids, while membrane separation is used for water-soluble acidic and bola-type sophorolipids.
In summary, sophorolipid separation methods are diverse but mostly for small-scale. The lack of low-energy, large-scale precision separation equipment is a major barrier to sophorolipid large-scale commercialization.
Main Applications
Petroleum Industry
Many surfactants enhance oil recovery, with microbial surfactants being highly promising. Studies show biosurfactants’ oil displacement efficiency is several times higher than synthetic ones. Using sophorolipids alone can improve oil recovery, and compounding with polysaccharides or rhamnolipids makes it even more effective.
Daily Chemical Industry
Sophorolipids are mild and can inhibit microbial activity, suitable for cosmetics. France’s Soliance launched a cosmetic with sophorolipids as the main ingredient, effectively inhibiting Propionibacterium acnes and Corynebacterium, while suppressing lipase activity to prevent sebum breakdown into free fatty acids.
Agriculture and Animal Husbandry
Crops use large amounts of pesticides, mostly lipophilic, requiring emulsifiers for stability and spraying. Sophorolipids offer excellent emulsification, replacing non-degradable chemical surfactants in pesticides. Their unique plant cell affinity enhances pesticide efficiency.
Environmental Protection
Like rhamnolipids, sophorolipids degrade certain metal ions and hydrocarbons. They can also separate asphalt from tar.
Nanotechnology
Sophorolipids combined with metal nanoparticles: Nanoparticles are “capped” to prevent aggregation and ensure uniform dispersion in organic solvents or water, gaining hydrophilicity for easy dispersion. These sophorolipid-capped nanoparticles serve as carriers for many bioactive molecules.
Future Outlook
In recent years, numerous reports on sophorolipid research have emerged. Currently, sophorolipids hold broad prospects in petroleum and daily chemical industries, with high-purity products priced at up to $30,000 per ton, far exceeding conventional surfactants. By developing high-density cell culture processes, improving fermentation levels, and reducing separation costs, the competitiveness of sophorolipids will greatly increase.
For premium sophorolipids and other microbial surfactants, explore Book Chem’s offerings at Sophorolipid Products. As innovators in bio-based surfactants, Book Chem is dedicated to advancing sophorolipids for sustainable solutions.
Summary of Surfactant Knowledge Points
A Series for Easy Understanding
Green surfactants
- Classification and Properties of Green Surfactants
- Natural Polysaccharide-Based Green Surfactants
- Properties and Applications of Anionic Amino Acid Surfactants
- Synthesis Processes of Anionic Amino Acid Surfactants
- Microbial Surfactants: Sophorolipids
- Types and Structures of Cardanol-Based Surfactants
New surfactants
- Synthesis and Applications of Phosphate Ester Surfactants
- Gemini Surfactants and Their Antibacterial Properties
- Low-Foam Surfactants: Types, Properties, and Applications
- Impact of Polyether Structure on Product Performance
- Types and Applications of Isomeric Alcohol Special Surfactants
- Research Progress on Secondary Alcohol Ethoxylates

