In cosmetic cream and lotion development, fatty alcohol selection is far more than a thickener choice. Many formulators face puzzling issues: why does swapping one fatty alcohol cause drastic viscosity changes? Why replacing C16/18 with high-melting pure C18 stearyl alcohol for better heat resistance results in failed texture or separation? The answer lies in fatty alcohols as the backbone of gel networks (lamellar liquid crystalline structures). Drawing from G.M. Eccleston’s landmark studies (IJCS 1982 & Colloids and Surfaces 1997), this guide reveals the hidden science and practical rules for optimal fatty alcohol performance.
Fatty Alcohols in Nonionic Emulsifier Systems
01、Myth-Busting: The Failure of Pure C18
Intuitively, pure stearyl alcohol (C18, m.p. ~58°C) should create thicker, more heat-resistant creams than cetyl alcohol (C16, m.p. ~49°C). Reality proves otherwise.
Eccleston’s 1982 study used PEG-1000 monostearate (common nonionic) with different fatty alcohols. After 30 days of viscosity tracking, results were striking:
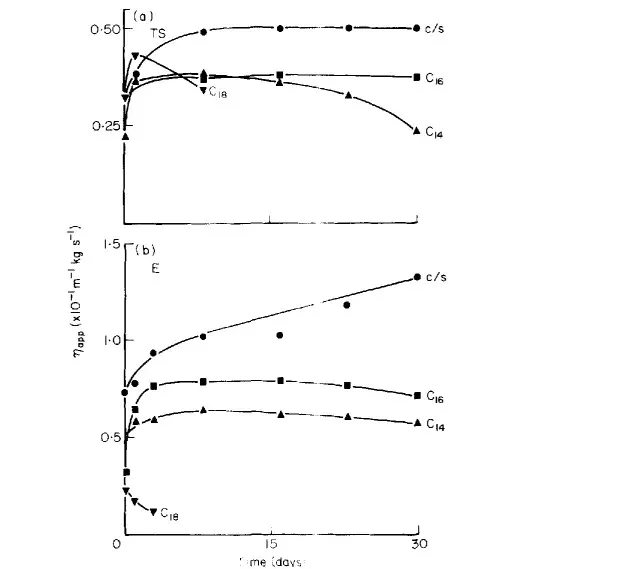
C16/18 blend steadily increased viscosity (stronger structure over time). Pure C18 remained extremely low—producing a mobile liquid prone to phase separation.
Table 1: Performance comparison in nonionic systems
- C16/18 Blend → Ideal semi-solid cream, best long-term stability
- Pure C16 → Softer cream, risk of thinning over time
- Pure C14 → Initial rise followed by drop, weak structure
- Pure C18 → Very low viscosity, no structure, rapid breaking
02、Core Mechanism: Swelling and Lamellar Gel Networks
Cream viscosity comes primarily from the continuous aqueous phase structure, not just oil droplet packing. When emulsifier + fatty alcohol + water interact, they form lamellar gel networks (layered structures) that trap large amounts of water—like a mille-feuille pastry.
This process is called swelling.
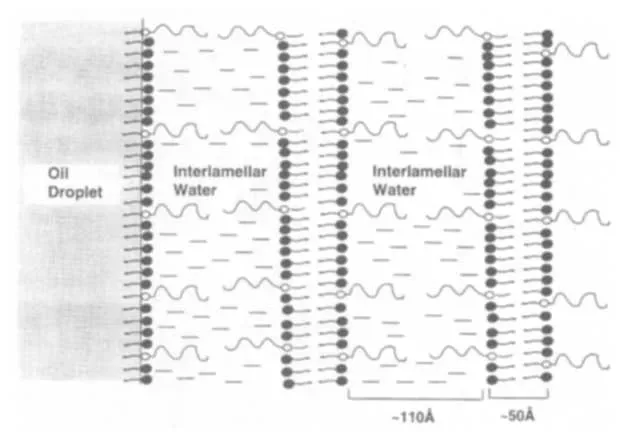
In nonionic systems (PEG esters, glucosides, A165, etc.), weak hydration forces limit penetration into tightly packed pure C18 crystals—no swelling, no framework.
Mixing C16 (shorter) with C18 (longer) introduces chain-length mismatch. The extra carbons in C18 protrude, preventing perfect alignment and creating micro-voids and lattice defects. Nonionic emulsifiers easily insert into these gaps, forming robust, water-trapping networks.
Table 2: Microscopic differences — Blend vs. Pure C18
- Crystal Order → Blend: Low (disordered) → Pure C18: High (tight)
- Emulsifier Penetration → Blend: Easy → Pure C18: Difficult
- α-Gel Formation → Blend: Yes (water-rich) → Pure C18: No (crystallizes out)
- Macro Result → Thick, smooth, stable → Thin, grainy, watery
03、Microscopic Evidence: Visual Proof of Interaction
Polarized light microscopy confirms the theory. Eccleston placed fatty alcohol crystals on slides, added emulsifier solution, and observed edge behavior over time.
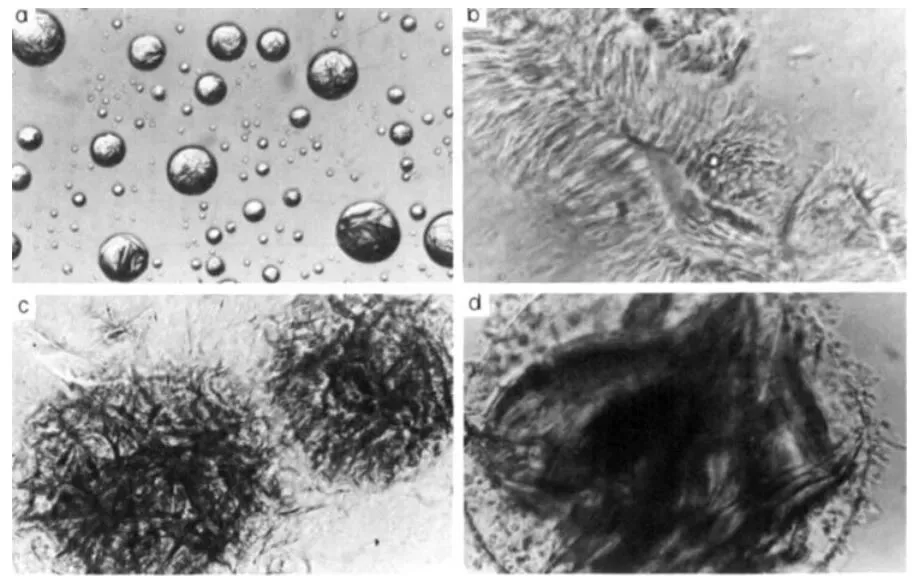
Figure 3:
- (a) Myristyl alcohol (C14) crystal
- (b) C14 + emulsifier (6 h) — rapid swelling
- (c) Cetyl alcohol (C16) + emulsifier (6 h) — gradual interaction
- (d) Stearyl alcohol (C18) + emulsifier (24 h) — sharp edges, minimal change
Pure C18 crystals remain distinct even after 24 hours—no meaningful interaction.
Table 3: Diffusion experiment summary (PEG-1000 MS system)
- C14 → Very fast (<6 h), extensive structure
- C16 → Moderate (~24 h), swelling occurs
- C18 → Extremely slow/none (>24 h), no effective gel
Practical Guide for Formulators
Based on Eccleston’s findings, follow these rules when using nonionic emulsifiers (PEG esters, glucosides, A165, etc.):
Table 4: Formulation recommendations
- Standard O/W Cream → Preferred: C16/18 cetyl-stearyl alcohol — highest tolerance, strongest network
- Heat Resistance → Avoid pure C18 swap. Add small % C22 behenyl alcohol or polymeric thickener
- Nonionic Systems → High risk with high-purity alcohols — blends are safer
- Texture Thinning → Perform microscopy to check swelling; batch variation in crystal habit may be culprit
Summary: Fatty alcohols are not mere fillers—their microscopic crystallization governs macroscopic fate. Mastering gel network theory elevates formulators from recipe makers to true scientists.
For more on related products, visit Surfactants. Questions? Contact us. In conclusion, smart fatty alcohol selection—especially C16/18 blends over pure stearyl alcohol—is critical for building stable, elegant gel network structures in modern cosmetic formulations.

